Multiple choice question for engineering
Set 1
1-10. A bucket contains 56 gms of C2H4, 36 gms of H2O and 44 gms of C3H8. (Molecular weights of the components are 28, 18 and 44 gms respectively)
1. How many moles of C2H4 are there in the bucket?
a) 1
b) 2
c) 4
d) 6
Answer
Answer: b [Reason:] Number of moles = mass / molecular weight.
2. How many moles of H2O are there in the bucket?
a) 1
b) 2
c) 4
d) 6
Answer
Answer: b [Reason:] Number of moles = mass / molecular weight.
3. How many moles of C3H8 are there in the bucket?
a) 1
b) 2
c) 4
d) 6
Answer
Answer: a [Reason:] Number of moles = mass / molecular weight.
4. What are the number moles of Oxgen present there in the bucket?
a) 1
b) 2
c) 4
d) 6
Answer
Answer: a [Reason:] Number of moles of Oxygen = 1/2(Number of moles of H2O) .
5. How many moles of H2 are there in the bucket?
a) 3
b) 6
c) 10
d) 12
Answer
Answer: c [Reason:] Moles of H2 = Moles of H2 in (2 Moles of C2H4+ 2 Moles of H2O + 1 Mole of C3H8).
6. What is mole percentage of C2H4?
a) 0.2
b) 0.4
c) 0.6
d) 0.8
Answer
Answer: b [Reason:] Mole percentage = Moles of the component / Total moles.
7. What is mole percentage of H2O?
a) 0.2
b) 0.4
c) 0.6
d) 0.8
Answer
Answer: b [Reason:] Mole percentage = Moles of the component / Total moles.
8. What is mole percentage of C3H8?
a) 0.2
b) 0.4
c) 0.6
d) 0.8
Answer
Answer: a [Reason:] Mole percentage = Moles of the component / Total moles.
9. What is the weight percentage of C2H4?
a) 41.17
b) 51.17
c) 61.17
d) 71.17
Answer
Answer: a [Reason:] Weight percentage = Weight of the component / Total weight.
10. What is the weight percentage of H2O?
a) 26.47
b) 36.47
c) 47.47
d) 58.47
Answer
Answer: a [Reason:] Weight percentage = Weight of the component / Total weight.
Set 2
1. Distillation is a separating process based on __________ of liquid mixture.
a) Vaporizing
b) Condensation
c) Freezing
d) None of the mentioned
Answer
Answer: a [Reason:] . Distillation is a separating process based on Vaporizing of liquid mixture.
2-8. For the given distillation column
1000 Kg of feed is given to the column and the efficiency of the column is 10%.
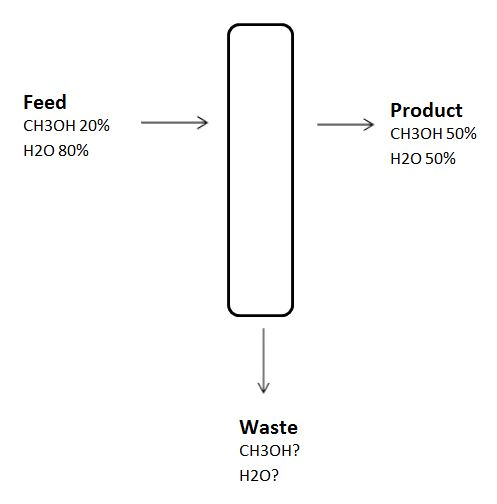
2. What is the amount of product?
a) 10 Kg
b) 100 Kg
c) 1000 Kg
d) None of the mentioned
Answer
Answer: b [Reason:] Product in Kg = 1000*0.1 = 100 Kg.
3. What is amount of CH3OH in the feed?
a) 200 Kg
b) 400 Kg
c) 600 Kg
d) 800 Kg
Answer
Answer: a [Reason:] CH3OH in the feed = 1000*0.2 = 200 Kg.
4. What is amount of H2O in the feed?
a) 200 Kg
b) 400 Kg
c) 600 Kg
d) 800 Kg
Answer
Answer: d [Reason:] H2O in the feed = 1000*0.8 = 800 Kg.
5. What is amount of CH3OH in the product?
a) 25 Kg
b) 50 Kg
c) 75 Kg
d) 100 Kg
Answer
Answer: b [Reason:] CH3OH in the product = 100*0.5 = 500 Kg.
6. What is amount of H2O in the product?
a) 25 Kg
b) 50 Kg
c) 75 Kg
d) 100 Kg
Answer
Answer: [Reason:] H2O in the product = 100*0.5 = 500 Kg.
7. What is amount of CH3OH in the waste?
a) 150 Kg
b) 250 Kg
c) 450 Kg
d) 750 Kg
Answer
Answer: a [Reason:] Waste = Product – Feed.
8. What is amount of H2O in the waste?
a) 150 Kg
b) 250 Kg
c) 450 Kg
d) 750 Kg
Answer
Answer: [Reason:] Waste = Product – Feed.
9. 50 Kg of a solid liquid mixture containing 10% solid and 90% water is left open in atmosphere, after some time the water is 80%, what is the weight of the mixture now?
a) 10 Kg
b) 25 Kg
c) 35 Kg
d) 50 Kg
Answer
Answer: b [Reason:] Make solid and water balance.
10. How much water was evaporated?
a) 10 Kg
b) 25 Kg
c) 35 Kg
d) 50 Kg
Answer
Answer: b [Reason:] Evaporated water = Initial amount of solution – Final amount of solution.
Set 3
1. A ____________ stream is one that skips one or more stages of the process and goes directly to another downstream stage.
a) Recycle
b) Purge
c) Bypass
d) None of the mentioned
Answer
Answer: c
Explanation: A Bypass stream is one that skips one or more stages of the process and goes directly to another downstream stage.
2. A ___________ stream is a stream bled off from the process to remove an accumulation of inerts or unwanted material.
a) Recycle
b) Purge
c) Bypass
d) None of the mentioned
Answer
Answer: b [Reason:] A Purge stream is a stream bled off from the process to remove an accumulation of inerts or unwanted material.
3-4. For the given process
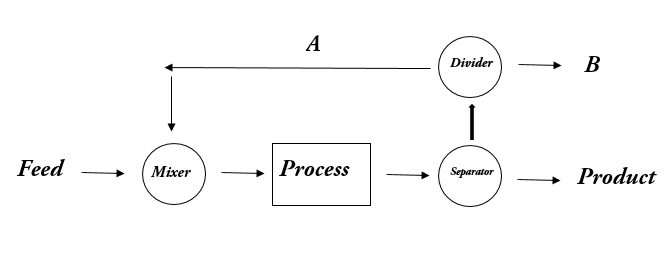
3. What is A in the process?
a) Recycle
b) Purge
c) Bypass
d) None of the mentioned
Answer
Answer: a [Reason:] Material that leaves a process unit that is the downstream and is returned to the same unit is called recycle.
4. What is B in the process?
a) Recycle
b) Purge
c) Bypass
d) None of the mentioned
Answer
Answer: b [Reason:] A Purge stream is a stream bled off from the process to remove an accumulation of inerts or unwanted material.
5. For the given process, A is
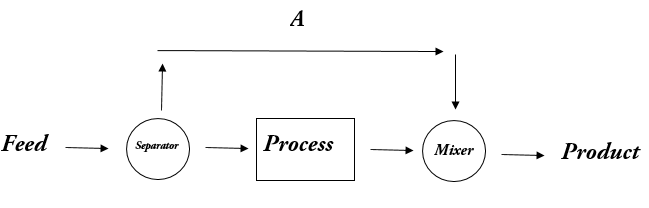
a) Recycle
b) Purge
c) Bypass
d) None of the mentioned
Answer
Answer: c [Reason:] A Bypass stream is one that skips one or more stages of the process and goes directly to another downstream stage.
6. A bypass stream does pass through __________
a) Mixer
b) Process
c) Separator
d) None of the mentioned
Answer
Answer: b [Reason:] A bypass stream directly goes from divider to separator skipping the process.
7. For the given process, how many recycle streams are there
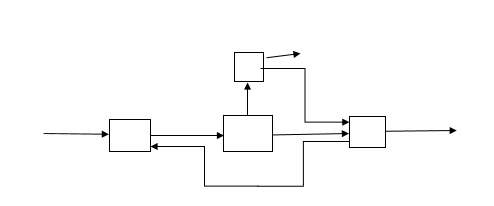
a) 1
b) 2
c) 3
d) 4
Answer
Answer: a [Reason:] Material that leaves a process unit that is the downstream and is returned to the same unit is called recycle.
8. A bypass stream can be used to control the composition of a final exit stream. The statement is
a) True
b) False
c) Always False
d) None of the mentioned
Answer
Answer: a [Reason:] A bypass stream can be used to control the composition of a final exit stream from a unit by mixing the bypass stream and the unit exit stream in suitable proportion to obtain the desired final composition.
9. Purge stream comes from?
a) Feed stream
b) Product stream
c) Recycle stream
d) None of the mentioned
Answer
Answer: c [Reason:] Purge stream is the removal of unwanted material from the recycle stream.
10. Process simulators are used for the activities like process design, process analysis.
The above given statement is
a) Correct
b) Incorrect
c) Depends on the process
d) None of the mentioned
Answer
Answer: a [Reason:] Process simulation is a model-based representation of chemical, physical, biological, and other technical processes and unit operations in software.
Set 4
1. A ____________ stream is one that skips one or more stages of the process and goes directly to another downstream stage.
a) Recycle
b) Purge
c) Bypass
d) None of the mentioned
Answer
Answer: c
Explanation: A Bypass stream is one that skips one or more stages of the process and goes directly to another downstream stage.
2. A ___________ stream is a stream bled off from the process to remove an accumulation of inerts or unwanted material.
a) Recycle
b) Purge
c) Bypass
d) None of the mentioned
Answer
Answer: b [Reason:] A Purge stream is a stream bled off from the process to remove an accumulation of inerts or unwanted material.
3-4. For the given process

3. What is A in the process?
a) Recycle
b) Purge
c) Bypass
d) None of the mentioned
Answer
Answer: a [Reason:] Material that leaves a process unit that is the downstream and is returned to the same unit is called recycle.
4. What is B in the process?
a) Recycle
b) Purge
c) Bypass
d) None of the mentioned
Answer
Answer: b [Reason:] A Purge stream is a stream bled off from the process to remove an accumulation of inerts or unwanted material.
5. For the given process, A is

a) Recycle
b) Purge
c) Bypass
d) None of the mentioned
Answer
Answer: c [Reason:] A Bypass stream is one that skips one or more stages of the process and goes directly to another downstream stage.
6. A bypass stream does pass through __________
a) Mixer
b) Process
c) Separator
d) None of the mentioned
Answer
Answer: b [Reason:] A bypass stream directly goes from divider to separator skipping the process.
7. For the given process, how many recycle streams are there

a) 1
b) 2
c) 3
d) 4
Answer
Answer: a [Reason:] Material that leaves a process unit that is the downstream and is returned to the same unit is called recycle.
8. A bypass stream can be used to control the composition of a final exit stream. The statement is
a) True
b) False
c) Always False
d) None of the mentioned
Answer
Answer: a [Reason:] A bypass stream can be used to control the composition of a final exit stream from a unit by mixing the bypass stream and the unit exit stream in suitable proportion to obtain the desired final composition.
9. Purge stream comes from?
a) Feed stream
b) Product stream
c) Recycle stream
d) None of the mentioned
Answer
Answer: c [Reason:] Purge stream is the removal of unwanted material from the recycle stream.
10. Process simulators are used for the activities like process design, process analysis.
The above given statement is
a) Correct
b) Incorrect
c) Depends on the process
d) None of the mentioned
Answer
Answer: a [Reason:] Process simulation is a model-based representation of chemical, physical, biological, and other technical processes and unit operations in software.
Set 5
1-6. In Ammonia production, a reactor is supplied with a feed of 10 moles of N2¬ and 40 moles of H2. If the reaction takes place completely,
1. What is the extent of the reaction?
a) 10
b) 15
c) 20
d) None of the mentioned
Answer
Answer: a [Reason:] Extent of the reaction = Moles reacting in the reaction.
2. What is the number of moles of N2 after the reaction?
a) 0
b) 5
c) 10
d) None of the mentioned
Answer
Answer: a [Reason:] The reaction completely takes place, so the moles of NH3 are zero as it is a limiting reactant.
3. What is the number of moles of H2 after the reaction?
a) 10
b) 15
c) 20
d) None of the mentioned
Answer
Answer: a [Reason:] Moles of H2 = Initial moles of H2 – 3(Extent of the reaction).
4. What is the number of moles of NH3 after the reaction?
a) 10
b) 15
c) 20
d) None of the mentioned
Answer
Answer: c [Reason:] Moles of NH3 = 2(Extent of the reaction).
5. Which one is the limiting reactant?
a) N2
b) H2
c) Both N2 and H2
d) Neither N2 nor H2
Answer
Answer: a [Reason:] N2 has lower extent of reaction.
6. Which one is the excess reactant?
a) N2
b) H2
c) Both N2 and H2
d) Neither N2 nor H2
Answer
Answer: b [Reason:] H2 has higher extent of reaction.
7-10. For a given reaction reactor is supplied with a feed of composition 46% C3H7O4, 44% O2 and 10% N2 and if 50% of the limiting reagent conversion takes place.
4C3H7O4 + 11O2 → 12CO2 + 14H2O
7. What is the percentage of O2 in the product?
a) 10
b) 18
c) 38
d) 54
Answer
Answer: b [Reason:] Assume that 100 moles of feed is present in the reactor and do the material balance for each, Percentage of O2 = 18%.
8. What is the percentage of N2 in the product?
a) 8
b) 18
c) 34
d) 46
Answer
Answer: a [Reason:] Assume that 100 moles of feed is present in the reactor and do the material balance for each, Percentage of N2 = 8%.
9. What is the percentage of C3H7O4 in the product?
a) 10
b) 16
c) 31
d) 43
Answer
Answer: c [Reason:] Assume that 100 moles of feed is present in the reactor and do the material balance for each, Percentage of C3H7O4 = 31%.
10. What is the percentage of H2O in the product?
a) 15
b) 23
c) 39
d) 51
Answer
Answer: b [Reason:] Assume that 100 moles of feed is present in the reactor and do the material balance for each, Percentage of H2O = 23%.
