Multiple choice question for engineering
Set 1
1-2. A membrane is used for separation of gases from waste. If the feed stream contains 20% CO2 and 80% SO2 and the product contains 25% CO2 and 75% SO2. Assume that the waste stream amounts to 80% of the input stream.
1. What is the composition of CO2 in waste stream?
a) 0.1875
b) 1.1685
c) 0.1485
d) 0.1285
Answer
Answer: a [Reason:] Mole balances for both the gases.
2. What is the composition of SO2 in waste stream?
a) 0.8175
b) 0.8165
c) 0.8145
d) 0.8125
Answer
Answer: d [Reason:] Mole balances for both the gases.
3. Below given statements for distillation is
In distillation, a liquid mixture is boiled to produce a vapour of different composition that moves away from the liquid.
a) True
b) False
c) Partially correct
d) None of the mentioned
Answer
Answer: d [Reason:] In distillation, a liquid mixture is boiled to produce a vapour of different composition that moves away from the liquid.
4-5. In a distillation column for a feed of1000 kg the output is 80% of the feed and remaining 20% is waste. The feed contains 20% of Acetone and rest Water. The product contains 20% of Acetone and 40% of Water.
4. How much Acetone is there in waste?
a) 20 kg
b) 40 kg
c) 60 kg
d) 80 kg
Answer
Answer: b [Reason:] Mass balance for each component.
5. Moles of water in waste?
a) 4.45
b) 8.89
c) 16.67
d) 20.48
Answer
Answer: b [Reason:] Mass balance for each component.
6. Correct statement for a tie component in material balance is
a) Enters a process in only one stream
b) Leaves in only one stream
c) Does not react inside the process
d) All of the mentioned
Answer
Answer: d [Reason:] A tie component full-fills a, b, c all three conditions.
7-8. For the given drying process, a material (Called SRU) is dried and feed and outputs are as shown in the figure.
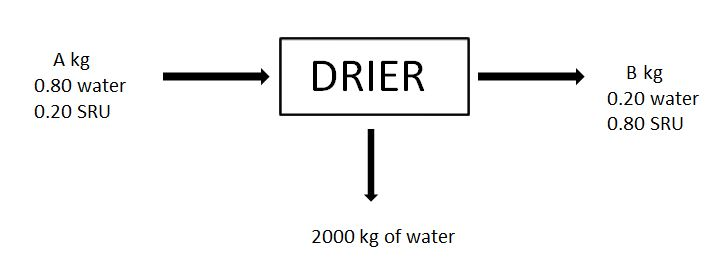
7. A, in kg is
a) 1666.67
b) 2666.67
c) 3666.67
d) 4666.67
Answer
Answer: b [Reason:] Mass balance for each component.
8. B, in kg is
a) 666.67
b) 566.67
c) 466.67
d) 366.67
Answer
Answer: a [Reason:] Mass balance for each component.
9-10. A batch of 20% of Acetic acid is prepared by mixing the Acetic acid of two containers A and B. A (400 kg) contains 80% of Acetic acidand B contains 15% of Acetic acid.
9. How much kg of acid is prepared?
a) 5700
b) 7600
c) 8000
d) 9800
Answer
Answer: b [Reason:] Mass balance for each component.
10. How much B in kg is used?
a) 4000
b) 6000
c) 8000
d) 10000
Answer
Answer: c [Reason:] Mass balance for each component.
Set 2
1-8. In a reactor, the feed components are NaOH, H2O and HCl. The mass fractions of these components in feed are 0.3, 0.5, 0.2 respectively. The total amount of feed is 800 Kg.
At the product side, the total amount of product is 400 Kg and amount of H2O collected is 300 Kg. The mass ratio of NaOH and HCl is 0.6.
Assume that there is no reaction is occurring in the reaction.
1. What is the amount of NaOH present in the feed?
a) 240 Kg
b) 480 Kg
c) 560 Kg
d) 800 Kg
Answer
Answer: a [Reason:] Mass fraction = Mass of the interested component / Total mass.
2. What is the amount of HCl present in the feed?
a) 80 Kg
b) 160 Kg
c) 240 Kg
d) 320 Kg
Answer
Answer: b [Reason:] Mass fraction = Mass of the interested component / Total mass.
3. What is the amount of H2O present in the feed?
a) 100 Kg
b) 200 Kg
c) 400 Kg
d) 800 Kg
Answer
Answer: c [Reason:] Mass fraction = Mass of the interested component / Total mass.
4. What is the amount of NaOH present in the product?
a) 21.5 Kg
b) 31.5 Kg
c) 37.5 Kg
d) 40.5 Kg
Answer
Answer: c [Reason:] Mass balance for NaOH.
5. What is the amount of HCl present in the product?
a) 62.5 Kg
b) 56.5 Kg
c) 40.5 Kg
d) 32.5 Kg
Answer
Answer: a [Reason:] Mass balance for HCl.
6. How many Kgs of NaOH is there in the reactor?
a) 101.5 Kg
b) 202.5 Kg
c) 303.5 Kg
d) 404.5 Kg
Answer
Answer: b [Reason:] Mass balance for NaOH.
7. How many Kgs of HCl is there in the reactor?
a) 111.5 Kg
b) 122.5 Kg
c) 133.5 Kg
d) 144.5 Kg
Answer
Answer: b [Reason:] Mass balance for HCl.
8. How many Kgs of H2O is there in the reactor?
a) 100 Kg
b) 200 Kg
c) 300 Kg
d) 400 Kg
Answer
Answer: a [Reason:] Mass balance for H2O.
9. An aqueous solution with NaOH 10 g/L at the rate is 100 L/min and an organic compound with no NaOH at the rate 50 L/min were put into an extraction machine and produced aqueous solution with NaOH 1 g/L, what is the amount of NaOH in organic compound after extraction?
a) 2 g/L
b) 5 g/L
c) 9 g/L
d) 15 g/L
Answer
Answer: c [Reason:] NaOH balance equation, 100(10) + 50(0) = 100(1) + 50(x).
10. To obtain a 100 Kg of 40 % N2 solution, how much nitrogen must be added to a 20% nitrogen solution?
a) 10 Kg
b) 25 Kg
c) 50 Kg
d) 75 Kg
Answer
Answer: b [Reason:] Use Both nitrogen and solvent balance.
Set 3
1-8. In a reactor, the feed components are NaOH, H2O and HCl. The mass fractions of these components in feed are 0.3, 0.5, 0.2 respectively. The total amount of feed is 800 Kg.
At the product side, the total amount of product is 400 Kg and amount of H2O collected is 300 Kg. The mass ratio of NaOH and HCl is 0.6.
Assume that there is no reaction is occurring in the reaction.
1. What is the amount of NaOH present in the feed?
a) 240 Kg
b) 480 Kg
c) 560 Kg
d) 800 Kg
Answer
Answer: a [Reason:] Mass fraction = Mass of the interested component / Total mass.
2. What is the amount of HCl present in the feed?
a) 80 Kg
b) 160 Kg
c) 240 Kg
d) 320 Kg
Answer
Answer: b [Reason:] Mass fraction = Mass of the interested component / Total mass.
3. What is the amount of H2O present in the feed?
a) 100 Kg
b) 200 Kg
c) 400 Kg
d) 800 Kg
Answer
Answer: c [Reason:] Mass fraction = Mass of the interested component / Total mass.
4. What is the amount of NaOH present in the product?
a) 21.5 Kg
b) 31.5 Kg
c) 37.5 Kg
d) 40.5 Kg
Answer
Answer: c [Reason:] Mass balance for NaOH.
5. What is the amount of HCl present in the product?
a) 62.5 Kg
b) 56.5 Kg
c) 40.5 Kg
d) 32.5 Kg
Answer
Answer: a [Reason:] Mass balance for HCl.
6. How many Kgs of NaOH is there in the reactor?
a) 101.5 Kg
b) 202.5 Kg
c) 303.5 Kg
d) 404.5 Kg
Answer
Answer: b [Reason:] Mass balance for NaOH.
7. How many Kgs of HCl is there in the reactor?
a) 111.5 Kg
b) 122.5 Kg
c) 133.5 Kg
d) 144.5 Kg
Answer
Answer: b [Reason:] Mass balance for HCl.
8. How many Kgs of H2O is there in the reactor?
a) 100 Kg
b) 200 Kg
c) 300 Kg
d) 400 Kg
Answer
Answer: a [Reason:] Mass balance for H2O.
9. An aqueous solution with NaOH 10 g/L at the rate is 100 L/min and an organic compound with no NaOH at the rate 50 L/min were put into an extraction machine and produced aqueous solution with NaOH 1 g/L, what is the amount of NaOH in organic compound after extraction?
a) 2 g/L
b) 5 g/L
c) 9 g/L
d) 15 g/L
Answer
Answer: c [Reason:] NaOH balance equation, 100(10) + 50(0) = 100(1) + 50(x).
10. To obtain a 100 Kg of 40 % N2 solution, how much nitrogen must be added to a 20% nitrogen solution?
a) 10 Kg
b) 25 Kg
c) 50 Kg
d) 75 Kg
Answer
Answer: b [Reason:] Use Both nitrogen and solvent balance.
Set 4
1-8. In a reactor, the feed components are NaOH, H2O and HCl. The mass fractions of these components in feed are 0.3, 0.5, 0.2 respectively. The total amount of feed is 800 Kg.
At the product side, the total amount of product is 400 Kg and amount of H2O collected is 300 Kg. The mass ratio of NaOH and HCl is 0.6.
Assume that there is no reaction is occurring in the reaction.
1. What is the amount of NaOH present in the feed?
a) 240 Kg
b) 480 Kg
c) 560 Kg
d) 800 Kg
Answer
Answer: a [Reason:] Mass fraction = Mass of the interested component / Total mass.
2. What is the amount of HCl present in the feed?
a) 80 Kg
b) 160 Kg
c) 240 Kg
d) 320 Kg
Answer
Answer: b [Reason:] Mass fraction = Mass of the interested component / Total mass.
3. What is the amount of H2O present in the feed?
a) 100 Kg
b) 200 Kg
c) 400 Kg
d) 800 Kg
Answer
Answer: c [Reason:] Mass fraction = Mass of the interested component / Total mass.
4. What is the amount of NaOH present in the product?
a) 21.5 Kg
b) 31.5 Kg
c) 37.5 Kg
d) 40.5 Kg
Answer
Answer: c [Reason:] Mass balance for NaOH.
5. What is the amount of HCl present in the product?
a) 62.5 Kg
b) 56.5 Kg
c) 40.5 Kg
d) 32.5 Kg
Answer
Answer: a [Reason:] Mass balance for HCl.
6. How many Kgs of NaOH is there in the reactor?
a) 101.5 Kg
b) 202.5 Kg
c) 303.5 Kg
d) 404.5 Kg
Answer
Answer: b [Reason:] Mass balance for NaOH.
7. How many Kgs of HCl is there in the reactor?
a) 111.5 Kg
b) 122.5 Kg
c) 133.5 Kg
d) 144.5 Kg
Answer
Answer: b [Reason:] Mass balance for HCl.
8. How many Kgs of H2O is there in the reactor?
a) 100 Kg
b) 200 Kg
c) 300 Kg
d) 400 Kg
Answer
Answer: a [Reason:] Mass balance for H2O.
9. An aqueous solution with NaOH 10 g/L at the rate is 100 L/min and an organic compound with no NaOH at the rate 50 L/min were put into an extraction machine and produced aqueous solution with NaOH 1 g/L, what is the amount of NaOH in organic compound after extraction?
a) 2 g/L
b) 5 g/L
c) 9 g/L
d) 15 g/L
Answer
Answer: c [Reason:] NaOH balance equation, 100(10) + 50(0) = 100(1) + 50(x).
10. To obtain a 100 Kg of 40 % N2 solution, how much nitrogen must be added to a 20% nitrogen solution?
a) 10 Kg
b) 25 Kg
c) 50 Kg
d) 75 Kg
Answer
Answer: b [Reason:] Use Both nitrogen and solvent balance.
Set 5
1-8. In a reactor, the feed components are NaOH, H2O and HCl. The mass fractions of these components in feed are 0.3, 0.5, 0.2 respectively. The total amount of feed is 800 Kg.
At the product side, the total amount of product is 400 Kg and amount of H2O collected is 300 Kg. The mass ratio of NaOH and HCl is 0.6.
Assume that there is no reaction is occurring in the reaction.
1. What is the amount of NaOH present in the feed?
a) 240 Kg
b) 480 Kg
c) 560 Kg
d) 800 Kg
Answer
Answer: a [Reason:] Mass fraction = Mass of the interested component / Total mass.
2. What is the amount of HCl present in the feed?
a) 80 Kg
b) 160 Kg
c) 240 Kg
d) 320 Kg
Answer
Answer: b [Reason:] Mass fraction = Mass of the interested component / Total mass.
3. What is the amount of H2O present in the feed?
a) 100 Kg
b) 200 Kg
c) 400 Kg
d) 800 Kg
Answer
Answer: c [Reason:] Mass fraction = Mass of the interested component / Total mass.
4. What is the amount of NaOH present in the product?
a) 21.5 Kg
b) 31.5 Kg
c) 37.5 Kg
d) 40.5 Kg
Answer
Answer: c [Reason:] Mass balance for NaOH.
5. What is the amount of HCl present in the product?
a) 62.5 Kg
b) 56.5 Kg
c) 40.5 Kg
d) 32.5 Kg
Answer
Answer: a [Reason:] Mass balance for HCl.
6. How many Kgs of NaOH is there in the reactor?
a) 101.5 Kg
b) 202.5 Kg
c) 303.5 Kg
d) 404.5 Kg
Answer
Answer: b [Reason:] Mass balance for NaOH.
7. How many Kgs of HCl is there in the reactor?
a) 111.5 Kg
b) 122.5 Kg
c) 133.5 Kg
d) 144.5 Kg
Answer
Answer: b [Reason:] Mass balance for HCl.
8. How many Kgs of H2O is there in the reactor?
a) 100 Kg
b) 200 Kg
c) 300 Kg
d) 400 Kg
Answer
Answer: a [Reason:] Mass balance for H2O.
9. An aqueous solution with NaOH 10 g/L at the rate is 100 L/min and an organic compound with no NaOH at the rate 50 L/min were put into an extraction machine and produced aqueous solution with NaOH 1 g/L, what is the amount of NaOH in organic compound after extraction?
a) 2 g/L
b) 5 g/L
c) 9 g/L
d) 15 g/L
Answer
Answer: c [Reason:] NaOH balance equation, 100(10) + 50(0) = 100(1) + 50(x).
10. To obtain a 100 Kg of 40 % N2 solution, how much nitrogen must be added to a 20% nitrogen solution?
a) 10 Kg
b) 25 Kg
c) 50 Kg
d) 75 Kg
Answer
Answer: b [Reason:] Use Both nitrogen and solvent balance.
