Multiple choice question for engineering
Set 1
1- 4. For the given problem
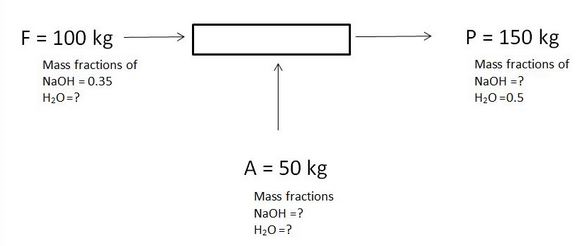
1. What is the mass fraction for H2O in feed and NaOH in product?
a) 0.50 and 0.50
b) 0.65 and 0.50
c) 0.65 and 0.65
d) 0.50 and 0.95
Answer
Answer: b [Reason:] ΣωI = 1.
2. What is the mass in kg of H2O in feed and NaOH in product?
a) 65 and 50
b) 95 and 50
c) 65 and 75
d) 50 and 50
Answer
Answer: c [Reason:] ΣωI = 1 and mi= ωIW.
3. What is the mass fraction of NaOH in A?
a) 0.2
b) 0.4
c) 0.6
d) 0.8
Answer
Answer: d [Reason:] ΣωI = 1 and ΣωIWi = 0 for each component.
4. What is the mass fraction of H2O in A?
a) 0.2
b) 0.4
c) 0.6
d) 0.8
Answer
Answer: a [Reason:] ΣωI = 1 and ΣωIWi = 0 for each component.
5-7. Tank A contains 90% O2 that is mixed with another tank B containing 30% of O2 to get a Tank C containing 65% O2 .
5. What is the ratio of the gas used from Tank A to that used from Tank B?
a) 1.0
b) 1.2
c) 1.4
d) 1.6
Answer
Answer: c [Reason:] Σxi = 1.
6. If A = 100 moles, what are the number of moles of B?
a) 51.4
b) 61.4
c) 71.4
d) 81.6
Answer
Answer: c [Reason:] Σxi = 1.
7. If A = 100 moles, what are the number of moles of C?
a) 151.4
b) 161.4
c) 171.4
d) 181.6
Answer
Answer: c [Reason:] Σxi = 1.
8. The number of variables whose values are unknown minus the number of independent equation is degree of freedom,
The above given statement is
a) True
b) False
c) It is not the definition of degrees of freedom
d) None of the mentioned
Answer
Answer: a [Reason:] The number of variables whose values are unknown minus the number of independent equation is degree of freedom.
9. Concept of material balance based upon?
a) Conservation of mass
b) Conservation of energy
c) Conservation of momentum
d) Conservation of Volume
Answer
Answer: a [Reason:] Material balance is based on conservation of mass.
10. Closed systems have ________ type of boundary wall.
a) Impermeable
b) Permeable
c) Rigid
d) None of the mentioned
Answer
Answer: a [Reason:] The matter is not allowed to cross the boundary wall of a closed system.
Set 2
1- 4. For the given problem

1. What is the mass fraction for H2O in feed and NaOH in product?
a) 0.50 and 0.50
b) 0.65 and 0.50
c) 0.65 and 0.65
d) 0.50 and 0.95
Answer
Answer: b [Reason:] ΣωI = 1.
2. What is the mass in kg of H2O in feed and NaOH in product?
a) 65 and 50
b) 95 and 50
c) 65 and 75
d) 50 and 50
Answer
Answer: c [Reason:] ΣωI = 1 and mi= ωIW.
3. What is the mass fraction of NaOH in A?
a) 0.2
b) 0.4
c) 0.6
d) 0.8
Answer
Answer: d [Reason:] ΣωI = 1 and ΣωIWi = 0 for each component.
4. What is the mass fraction of H2O in A?
a) 0.2
b) 0.4
c) 0.6
d) 0.8
Answer
Answer: a [Reason:] ΣωI = 1 and ΣωIWi = 0 for each component.
5-7. Tank A contains 90% O2 that is mixed with another tank B containing 30% of O2 to get a Tank C containing 65% O2 .
5. What is the ratio of the gas used from Tank A to that used from Tank B?
a) 1.0
b) 1.2
c) 1.4
d) 1.6
Answer
Answer: c [Reason:] Σxi = 1.
6. If A = 100 moles, what are the number of moles of B?
a) 51.4
b) 61.4
c) 71.4
d) 81.6
Answer
Answer: c [Reason:] Σxi = 1.
7. If A = 100 moles, what are the number of moles of C?
a) 151.4
b) 161.4
c) 171.4
d) 181.6
Answer
Answer: c [Reason:] Σxi = 1.
8. The number of variables whose values are unknown minus the number of independent equation is degree of freedom,
The above given statement is
a) True
b) False
c) It is not the definition of degrees of freedom
d) None of the mentioned
Answer
Answer: a [Reason:] The number of variables whose values are unknown minus the number of independent equation is degree of freedom.
9. Concept of material balance based upon?
a) Conservation of mass
b) Conservation of energy
c) Conservation of momentum
d) Conservation of Volume
Answer
Answer: a [Reason:] Material balance is based on conservation of mass.
10. Closed systems have ________ type of boundary wall.
a) Impermeable
b) Permeable
c) Rigid
d) None of the mentioned
Answer
Answer: a [Reason:] The matter is not allowed to cross the boundary wall of a closed system.
Set 3
1. ________ combines two or more flow streams.
a) Mixer
b) Splitter
c) Separator
d) Adder
Answer
Answer: a [Reason:] Mixer combines two or more flow streams.
2. Apparatus that divides a flow in to two or more streams
a) Mixer
b) Splitter
c) Separator
d) Adder
Answer
Answer: b [Reason:] Splitter divides a flow in to two or more streams.
3. Apparatus that produces two or more streams of different composition from the fluid entering the apparatus is called
a) Mixer
b) Splitter
c) Separator
d) Adder
Answer
Answer: c [Reason:] Separator produces two or more streams of different composition from the fluid entering the apparatus.
4. Streams produced by a splitter have ________ composition as the feed.
a) Same
b) Different
c) Depends on the splitter
d) None of the mentioned.
Answer
Answer: a [Reason:] Streams produced by a splitter have same composition as the feed.
5. __________ is a sequence of boxes, circles and other shapes used to represent operational features of a process flowsheet.
a) Block Diagram
b) Process flow diagram
c) flow chart
d) None of the mentioned
Answer
Answer: a [Reason:] Block diagram is a sequence of boxes, circles and other shapes used to represent operational features of a process flow sheet.
6. A graphical representation of a process layout is called
a) Flow sheet
b) Flow diagram
c) Block diagram
d) None of the mentioned
Answer
Answer: a [Reason:] A graphical representation of a process layout is called Flow sheet.
7. Correct statement for a well-mixed system is
a) Material within the system has uniform composition
b) Material within the system doesn`t have uniform composition
c) Exit stream have the different composition as the material inside the system
d) None of the mentioned
Answer
Answer: a [Reason:] Material within the system have uniform composition.
8. The conversion of a reactant based on the amount of material that enters and leaves a reactor is
a) Once through fraction conversion
b) Overall fraction conversion
c) Percentage conversion
d) None of the mentioned
Answer
Answer: a [Reason:] The conversion of a reactant based on the amount of material that enters and leaves a reactors is called once through fraction conversion.
9. The conversion of a reactant in a process with recycle based on the fresh feed of the reactant and the overall products.
a) Once through fraction conversion
b) Overall fraction conversion
c) Percentage conversion
d) None of the mentioned
Answer
Answer: b [Reason:] The conversion of a reactant in a process with recycle based on the fresh feed of the reactant and the overall products overall fraction conversion.
10. A designated part of a system is ______
a) Connection
b) Subsystem
c) Reactor
d) Block Diagram
Answer
Answer: b [Reason:] A designated part of a system is called Subsystem.
Set 4
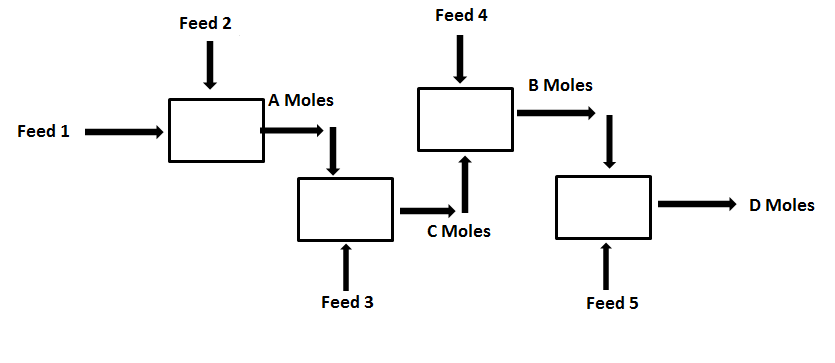
For the given process, If the values of Feed 1, 2, 3, 4, 5 are 1, 2, 3, 4, 5 moles respectively.
1. What is the value of A?
a) 1
b) 2
c) 3
d) 4
Answer
Answer: c [Reason:] A = Feed 1 + Feed 2.
2. What is the value of B?
a) 5
b) 10
c) 15
d) 20
Answer
Answer: b [Reason:] B = C + Feed 4.
3. What is the value of C?
a) 3
b) 6
c) 9
d) 12
Answer
Answer: b [Reason:] C = A + Feed 3.
4. What is the value of D?
a) 5
b) 10
c) 15
d) 20
Answer
Answer: c [Reason:] D = B + Feed 5.
5. If Feed 1 is not present here, what would be the value of D?
a) 5
b) 10
c) 14
d) 16
Answer
Answer: c [Reason:] D = Feed (2 + 3 + 4 + 5).
6. If Feed 5 is not present here, what would be the value of B?
a) 5
b) 10
c) 15
d) 20
Answer
Answer: b [Reason:] B is not depended on Feed 5.
7. If an addition feed of 2 moles are there near the feed 3, what is the value of A?
a) 1
b) 2
c) 3
d) 4
Answer
Answer: c [Reason:] A is not depended on the additional feed.
8. If an addition feed of 2 moles are there near the feed 3, what is the value of B?
a) 10
b) 12
c) 13
d) 14
Answer
Answer: b [Reason:] Now B = Initial B + 2.
9. If an addition feed of 2 moles are there near the feed 3, what is the value of C?
a) 6
b) 8
c) 10
d) 14
Answer
Answer: b [Reason:] Now C = Initial C + 2.
10. If an addition feed of 2 moles are there near the feed 3, what is the value of D?
a) 10
b) 12
c) 15
d) 17
Answer
Answer: d [Reason:] Now D = Initial D + 2.
Set 5
1-2. A membrane is used for separation of gases from waste. If the feed stream contains 20% CO2 and 80% SO2 and the product contains 25% CO2 and 75% SO2. Assume that the waste stream amounts to 80% of the input stream.
1. What is the composition of CO2 in waste stream?
a) 0.1875
b) 1.1685
c) 0.1485
d) 0.1285
Answer
Answer: a [Reason:] Mole balances for both the gases.
2. What is the composition of SO2 in waste stream?
a) 0.8175
b) 0.8165
c) 0.8145
d) 0.8125
Answer
Answer: d [Reason:] Mole balances for both the gases.
3. Below given statements for distillation is
In distillation, a liquid mixture is boiled to produce a vapour of different composition that moves away from the liquid.
a) True
b) False
c) Partially correct
d) None of the mentioned
Answer
Answer: d [Reason:] In distillation, a liquid mixture is boiled to produce a vapour of different composition that moves away from the liquid.
4-5. In a distillation column for a feed of1000 kg the output is 80% of the feed and remaining 20% is waste. The feed contains 20% of Acetone and rest Water. The product contains 20% of Acetone and 40% of Water.
4. How much Acetone is there in waste?
a) 20 kg
b) 40 kg
c) 60 kg
d) 80 kg
Answer
Answer: b [Reason:] Mass balance for each component.
5. Moles of water in waste?
a) 4.45
b) 8.89
c) 16.67
d) 20.48
Answer
Answer: b [Reason:] Mass balance for each component.
6. Correct statement for a tie component in material balance is
a) Enters a process in only one stream
b) Leaves in only one stream
c) Does not react inside the process
d) All of the mentioned
Answer
Answer: d [Reason:] A tie component full-fills a, b, c all three conditions.
7-8. For the given drying process, a material (Called SRU) is dried and feed and outputs are as shown in the figure.
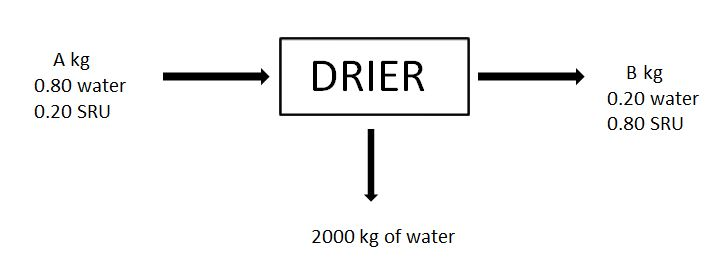
7. A, in kg is
a) 1666.67
b) 2666.67
c) 3666.67
d) 4666.67
Answer
Answer: b [Reason:] Mass balance for each component.
8. B, in kg is
a) 666.67
b) 566.67
c) 466.67
d) 366.67
Answer
Answer: a [Reason:] Mass balance for each component.
9-10. A batch of 20% of Acetic acid is prepared by mixing the Acetic acid of two containers A and B. A (400 kg) contains 80% of Acetic acidand B contains 15% of Acetic acid.
9. How much kg of acid is prepared?
a) 5700
b) 7600
c) 8000
d) 9800
Answer
Answer: b [Reason:] Mass balance for each component.
10. How much B in kg is used?
a) 4000
b) 6000
c) 8000
d) 10000
Answer
Answer: c [Reason:] Mass balance for each component.
