Multiple choice question for engineering
Set 1
1. Law of conservation of energy states that
a) Total energy of the universe is always constant
b) Total energy of the universe is always increasing
c) Total energy of the universe is always decreasing
d) None of the mentioned
Answer
Answer: a [Reason:] Law of conservation of energy states that total energy of the universe is always constant.
2. A mass having an initial kinetic energy of 40 Jules gets a potential energy of 10 Jules by reaching at a height h. What is the kinetic energy of the mass now?
a) 10 J
b) 20 J
c) 30 J
d) 40 J
Answer
Answer: b [Reason:] Law of conservation of energy states that total energy of the universe is always constant.
3-8. A block having a mass of 1 kg and velocity 2 m/s2 is at the ground.
3. What is the kinetic energy of the block?
a) 1 J
b) 2 J
c) 3 J
d) 4 J
Answer
Answer: b [Reason:] K.E. = ½ mv2.
4. If the block gets a height of 5 m, what is the potential energy of the block?
a) 49 J
b) 98 J
c) 87 J
d) 98 J
Answer
Answer: a [Reason:] P.E = mgh.
5. If the block gets a height of 0.2 m using some of the kinetic energy what is the kinetic energy of the block after reaching the extreme point?
a) 0.01 J
b) 0.02 J
c) 0.03 J
d) 0.04 J
Answer
Answer: d [Reason:] K.E. + P.E. = Total Energy = Constant.
6. The total energy of the block _________ in every operation.
a) Increases
b) Decreases
c) Remains constant
d) None of the mentioned
Answer
Answer: c [Reason:] The total energy of the block constant in every operation.
7. If the block uses the whole energy to gain some height, what will be that height?
a) 0.104 m
b) 0.204 m
c) 0.304 m
d) 0.404 m
Answer
Answer: b [Reason:] mgh = 2 J.
8. If the block is initially at a height of 2 m and with zero velocity. What is the maximum velocity it can gain?
a) 2.26
b) 4.26
c) 6.26
d) 8.26
Answer
Answer: c [Reason:] 1/2 mv2 = mgh.
9. If the velocity of the block is doubled, the kinetic energy of the block will be
a) Half
b) Double
c) Four times
d) None of the mentioned
Answer
Answer: c [Reason:] KE = ½ mv2.
10. If the height gained by the block is half of the initial case, the potential energy will also be ________ of the initial potential energy.
a) Half
b) Double
c) Four times
d) None of the mentioned
Answer
Answer: a [Reason:] PE = mgh.
Set 2
1. Enthalpy (H) is
a) H = U + PV
b) H = U – PV
c) H = U*PV
d) None of the mentioned
Where: U-Internal energy, P- Pressure, V- Volume
Answer
Answer: a [Reason:] H = U + PV.
2. Specific enthalpy is the function of
a) Temperature and Pressure
b) Temperature and Volume
c) Pressure and Volume
d) None of the mentioned
Answer
Answer: a [Reason:] H = f(T, P).
3. Cp is the change in enthalpy with respect to temperature at constant _____________
a) Pressure
b) Volume
c) Temperature
d) None of the mentioned
Answer
Answer: a [Reason:] Cp is the change in enthalpy with respect to temperature at constant Pressure.
4. For ideal gases, Enthalpy and Internal energy is only the function of __________
a) Pressure
b) Volume
c) Temperature
d) None of the mentioned
Answer
Answer: c [Reason:] For ideal gases, Enthalpy and Internal energy is only the function of Temperature.
5. Enthalpy has no absolute value. Only changes can be calculated.
The given statement is
a) Correct
b) Incorrect
c) Always incorrect
d) None of the mentioned
Answer
Answer: a [Reason:] Enthalpy is calculated with respect to a reference, there is no absolute value.
6. For ideal gases, the heat capacity relation is
a) Cp = Cv – R
b) Cv = Cp + R
c) Cp – Cv = R
d) None of the mentioned
Answer
Answer: c [Reason:] Cp – Cv = R.
7-10. For the given figure
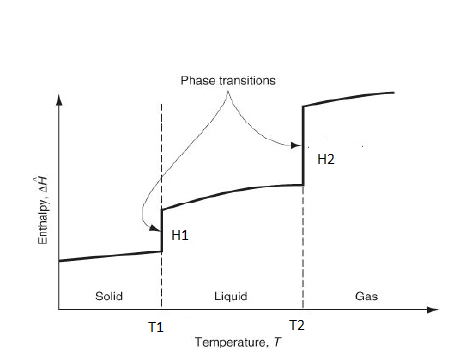
7. What is T1?
a) T, melting
b) T, freezing
c) T, vaporization
d) T, condensation
Answer
Answer: a [Reason:] T1 is the melting temperature.
8. What is T2?
a) T, melting
b) T, freezing
c) T, vaporization
d) T, condensation
Answer
Answer: c [Reason:] T2 is the vaporization temperature.
9. What is H1?
a) Heat of fusion
b) Heat of vaporization
c) Heat of sublimation
d) None of the mentioned
Answer
Answer: a [Reason:] H1 is the heat of fusion.
10. What is H2?
a) Heat of fusion
b) Heat of vaporization
c) Heat of sublimation
d) None of the mentioned
Answer
Answer: b [Reason:] H2 is the heat of vaporization.
Set 3
1. Enthalpy (H) is
a) H = U + PV
b) H = U – PV
c) H = U*PV
d) None of the mentioned
Where: U-Internal energy, P- Pressure, V- Volume
Answer
Answer: a [Reason:] H = U + PV.
2. Specific enthalpy is the function of
a) Temperature and Pressure
b) Temperature and Volume
c) Pressure and Volume
d) None of the mentioned
Answer
Answer: a [Reason:] H = f(T, P).
3. Cp is the change in enthalpy with respect to temperature at constant _____________
a) Pressure
b) Volume
c) Temperature
d) None of the mentioned
Answer
Answer: a [Reason:] Cp is the change in enthalpy with respect to temperature at constant Pressure.
4. For ideal gases, Enthalpy and Internal energy is only the function of __________
a) Pressure
b) Volume
c) Temperature
d) None of the mentioned
Answer
Answer: c [Reason:] For ideal gases, Enthalpy and Internal energy is only the function of Temperature.
5. Enthalpy has no absolute value. Only changes can be calculated.
The given statement is
a) Correct
b) Incorrect
c) Always incorrect
d) None of the mentioned
Answer
Answer: a [Reason:] Enthalpy is calculated with respect to a reference, there is no absolute value.
6. For ideal gases, the heat capacity relation is
a) Cp = Cv – R
b) Cv = Cp + R
c) Cp – Cv = R
d) None of the mentioned
Answer
Answer: c [Reason:] Cp – Cv = R.
7-10. For the given figure

7. What is T1?
a) T, melting
b) T, freezing
c) T, vaporization
d) T, condensation
Answer
Answer: a [Reason:] T1 is the melting temperature.
8. What is T2?
a) T, melting
b) T, freezing
c) T, vaporization
d) T, condensation
Answer
Answer: c [Reason:] T2 is the vaporization temperature.
9. What is H1?
a) Heat of fusion
b) Heat of vaporization
c) Heat of sublimation
d) None of the mentioned
Answer
Answer: a [Reason:] H1 is the heat of fusion.
10. What is H2?
a) Heat of fusion
b) Heat of vaporization
c) Heat of sublimation
d) None of the mentioned
Answer
Answer: b [Reason:] H2 is the heat of vaporization.
Set 4
1. What is the Enthalpy change for a 10 Kg mole Oxygen gas that is heated from 200 K to 400 K at constant pressure. (Cv = 40000 J/(Kg*mole)*(Degree Celsius))
a) 20*106
b) 40*106
c) 60*106
d) 80*106
Answer
Answer: d [Reason:] H2 – H1 = nCp(T2-T1).
2. What is the Enthalpy change for a 20 Kg mole Nitrogen gas that is heated from 100 K to 200 K at constant pressure. (Cv = 2000 J/(Kg*mole)*(Degree Celsius))
a) 2*106
b) 4*106
c) 6*106
d) 8*106
Answer
Answer: d [Reason:] H2 – H1 = nCp(T2-T1).
3. The negative of heat of vaporization is
a) Heat of Condensation
b) Heat of fusion
c) Heat of solidification
d) Heat of sublimation
Answer
Answer: a [Reason:] The negative of heat of vaporization is Heat of Condensation.
4. The Enthalpy change for the phase transition of melting is ___________
a) Heat of Condensation
b) Heat of fusion
c) Heat of solidification
d) Heat of sublimation
Answer
Answer: b [Reason:] The Enthalpy change for the phase transition of melting is the Heat of fusion.
5. The negative of the heat of fusion is
a) Heat of Condensation
b) Heat of vaporization
c) Heat of solidification
d) Heat of sublimation
Answer
Answer: c [Reason:] The negative of the heat of fusion is Heat of solidification.
6. The Enthalpy change of a solid directly to vapour is __________
a) Heat of Condensation
b) Heat of vaporization
c) Heat of solidification
d) Heat of sublimation
Answer
Answer: d [Reason:] The Enthalpy change of a solid directly to vapour is the Heat of sublimation.
7. The enthalpy change for the phase transition of a liquid to a vapour is
a) Heat of Condensation
b) Heat of vaporization
c) Heat of solidification
d) Heat of sublimation
Answer
Answer: b [Reason:] The enthalpy change for the phase transition of a liquid to a vapour is called Heat of vaporization.
8. An example of phase transition is
a) Liquid to gas phase
b) Gas to liquid phase
c) Solid to liquid phase
d) All of the mentioned
Answer
Answer: d [Reason:] Phase change is occurring in each case.
9. An enthalpy change that involve a phase transition is
a) Heat of the reaction
b) Latent Heat
c) Heat of fusion
d) Heat of sublimation
Answer
Answer: b [Reason:] An enthalpy change that involve a phase transition is Latent heat.
10. An enthalpy change that does not involve a phase transition is
a) Latent Heat
b) Sensible Heat
c) Heat of solidification
d) Heat of sublimation
Answer
Answer: b [Reason:] An enthalpy change that does not involve a phase transition is Sensible heat.
Set 5
1. What is the Enthalpy change for a 10 Kg mole Oxygen gas that is heated from 200 K to 400 K at constant pressure. (Cv = 40000 J/(Kg*mole)*(Degree Celsius))
a) 20*106
b) 40*106
c) 60*106
d) 80*106
Answer
Answer: d [Reason:] H2 – H1 = nCp(T2-T1).
2. What is the Enthalpy change for a 20 Kg mole Nitrogen gas that is heated from 100 K to 200 K at constant pressure. (Cv = 2000 J/(Kg*mole)*(Degree Celsius))
a) 2*106
b) 4*106
c) 6*106
d) 8*106
Answer
Answer: d [Reason:] H2 – H1 = nCp(T2-T1).
3. The negative of heat of vaporization is
a) Heat of Condensation
b) Heat of fusion
c) Heat of solidification
d) Heat of sublimation
Answer
Answer: a [Reason:] The negative of heat of vaporization is Heat of Condensation.
4. The Enthalpy change for the phase transition of melting is ___________
a) Heat of Condensation
b) Heat of fusion
c) Heat of solidification
d) Heat of sublimation
Answer
Answer: b [Reason:] The Enthalpy change for the phase transition of melting is the Heat of fusion.
5. The negative of the heat of fusion is
a) Heat of Condensation
b) Heat of vaporization
c) Heat of solidification
d) Heat of sublimation
Answer
Answer: c [Reason:] The negative of the heat of fusion is Heat of solidification.
6. The Enthalpy change of a solid directly to vapour is __________
a) Heat of Condensation
b) Heat of vaporization
c) Heat of solidification
d) Heat of sublimation
Answer
Answer: d [Reason:] The Enthalpy change of a solid directly to vapour is the Heat of sublimation.
7. The enthalpy change for the phase transition of a liquid to a vapour is
a) Heat of Condensation
b) Heat of vaporization
c) Heat of solidification
d) Heat of sublimation
Answer
Answer: b [Reason:] The enthalpy change for the phase transition of a liquid to a vapour is called Heat of vaporization.
8. An example of phase transition is
a) Liquid to gas phase
b) Gas to liquid phase
c) Solid to liquid phase
d) All of the mentioned
Answer
Answer: d [Reason:] Phase change is occurring in each case.
9. An enthalpy change that involve a phase transition is
a) Heat of the reaction
b) Latent Heat
c) Heat of fusion
d) Heat of sublimation
Answer
Answer: b [Reason:] An enthalpy change that involve a phase transition is Latent heat.
10. An enthalpy change that does not involve a phase transition is
a) Latent Heat
b) Sensible Heat
c) Heat of solidification
d) Heat of sublimation
Answer
Answer: b [Reason:] An enthalpy change that does not involve a phase transition is Sensible heat.
