Multiple choice question for engineering
Set 1
1. Mass per unit volume is called
a) Density
b) Molecular weight
c) Molar mass
d) Mole fraction
Answer
Answer: a [Reason:] Mass per unit volume is called density.
2. Densities for ________ and ________do not change significantly at ordinary conditions with pressure.
a) Gas, liquids
b) Liquids, solids
c) Solids, gas
d) None of the mentioned
Answer
Answer: b [Reason:] Densities for liquids and solids do not change significantly at ordinary conditions with pressure.
3. Specific volume is the inverse of
a) Volume
b) Pressure
c) Density
d) Flow rate
Answer
Answer: c [Reason:] Specific volume is the inverse of density.
4. Specific gravity is the ratio of density of a substance to the density of a reference material.
The above given statement is
a) True
b) False
c) Only for some specific materials
d) None of the mentioned
Answer
Answer: a [Reason:] Specific gravity is the ratio of density of a substance to the density of a reference material..
5. Density of water is
a) 1 gm/cm3
b) 1000 Kg/m3
c) 62.43 lb/ft3
d) All of the mentioned
Answer
Answer: a [Reason:] density of water = 1 gm/cm3 = 1000 Kg/m3 = 62.43 lb/ft3.
6. Specific gravity = 0.80 26˚/4˚ notation represents
a) Specific gravity when the solution is at 26˚C and the reference substance is at 4˚C is 0.80
b) Specific gravity when the solution is at 4˚C and the reference substance is at 26˚C is 0.80
c) Both a and b
d) Neither a nor b
Answer
Answer: a [Reason:] Simple rule for the representation.
7-8. Specific gravity of a liquid is 2.39.
7. What is the density of the liquid in g/cm3?
a) 2.39
b) 2.39102
c) 2.39×103
d) None of the mentioned
Answer
Answer: a [Reason:] Specific gravity is the ratio of the density of a substance to the density of a reference material.
8. What is the density of the liquid in kg/m3?
a) 2.39×103
b) 2.39
c) 2.39×102
d) None of the mentioned
Answer
Answer: a [Reason:] Specific gravity is the ratio of the density of a substance to the density of a reference material.
9. Specific gravity of a substance is 2.42. What is the density of the substance in lb/ft3?
a) 151
b) 252
c) 302
d) 402
Answer
Answer: a [Reason:] Specific gravity is the ratio of the density of a substance to the density of a reference material.
10. Mass that a cube contains is 40 gm and having side length of 1 cm. What is the density of the material inside the cube?
a) 40 gm/cm3
b) 20 gm/cm3
c) 10 gm/cm3
d) None of the mentioned
Answer
Answer: a [Reason:] Mass per unit volume is called density.
Set 2
1-3. A material having molecular weight 190, in the production of the given material the exit stream from the reactor containing water and the drug flows at a rate of 15.5 L/min. The material concentration is 32.2% in water. The specific gravity of the solution is 1.2.
1. What is density g/cm3 of the solution?
a) 1.2
b) 0.83
c) 12.4
d) 2.5
Answer
Answer: a [Reason:] Specific gravity is the ratio of the density of a material to the density of a reference material.
2. Concentration of the material in kg/L in the exit stream
a) 0.3864
b) 0.2033
c) 4.0673
d) 0.7728
Answer
Answer: a [Reason:] Take 1 min. basis and then using the flow rate of liquid, density of liquid and percentage of material calculate the concentration.
3. Flow rate of the material in kg/min.
a) 0.262
b) 0.252
c) 0.242
d) 0.232
Answer
Answer: a [Reason:] Take 1 min. basis and then using molecular weight of material, flow rate and percentage of material, calculate flow rate.
4. The density of material is 5 kg/cm3. What is its specific volume cm3/kg?
a) 0.2
b) 0.5
c) 5.0
d) 1.0
Answer
Answer: a [Reason:] Inverse of density is specific volume.
5. The specific weight is also known as ________
a) Specific density
b) Specific volume
c) Weight density
d) None of the mentioned
Answer
Answer: c [Reason:] The specific weight is also known as weight density.
6. The specific gravity of water is taken as
a) 0.01
b) 0.1
c) 1
d) 10
Answer
Answer: c [Reason:] While measuring the specific gravity of liquids, it is measured with respect to water.
7. Two statements are given as
I: Specific density is dimensionless.
II: When salt is added to water, density of the solution will increase.
Number of correct statement is
a) 0
b) 1
c) 2
d) None of the mentioned
Answer
Answer: c [Reason:] Specific density is the ratio of two densities and when salt is added to water, a high density material is added so finally the density rises .
8. An object has specific gravity greater than 1. When it is kept on the surface of water, the object would
a) Sink
b) Float
c) Half part of the object floats
d) None of the mentioned
Answer
Answer: a [Reason:] With respect to water, the specific gravity is greater than one so the density of the object is more than that of water.
9-10. A rectangular block measures 2 x 5 x 8 centimetres. Mass of the block is 66 grams.
9. Density of the block is?
a) 0.625 gm/cm3
b) 0.825 gm/cm3
c) 1.125 gm/cm3
d) 2.225 gm/cm3
Answer
Answer: b [Reason:] Density is the ratio of mass and volume of the block.
10. Specific gravity is?
a) 0.625
b) 0.825
c) 1.125
d) 2.225
Answer
Answer: b [Reason:] Specific density = 0.825/1 = 0.825, where the density of water is 1 gm/cm3.
Set 3
1-3. A material having molecular weight 190, in the production of the given material the exit stream from the reactor containing water and the drug flows at a rate of 15.5 L/min. The material concentration is 32.2% in water. The specific gravity of the solution is 1.2.
1. What is density g/cm3 of the solution?
a) 1.2
b) 0.83
c) 12.4
d) 2.5
Answer
Answer: a [Reason:] Specific gravity is the ratio of the density of a material to the density of a reference material.
2. Concentration of the material in kg/L in the exit stream
a) 0.3864
b) 0.2033
c) 4.0673
d) 0.7728
Answer
Answer: a [Reason:] Take 1 min. basis and then using the flow rate of liquid, density of liquid and percentage of material calculate the concentration.
3. Flow rate of the material in kg/min.
a) 0.262
b) 0.252
c) 0.242
d) 0.232
Answer
Answer: a [Reason:] Take 1 min. basis and then using molecular weight of material, flow rate and percentage of material, calculate flow rate.
4. The density of material is 5 kg/cm3. What is its specific volume cm3/kg?
a) 0.2
b) 0.5
c) 5.0
d) 1.0
Answer
Answer: a [Reason:] Inverse of density is specific volume.
5. The specific weight is also known as ________
a) Specific density
b) Specific volume
c) Weight density
d) None of the mentioned
Answer
Answer: c [Reason:] The specific weight is also known as weight density.
6. The specific gravity of water is taken as
a) 0.01
b) 0.1
c) 1
d) 10
Answer
Answer: c [Reason:] While measuring the specific gravity of liquids, it is measured with respect to water.
7. Two statements are given as
I: Specific density is dimensionless.
II: When salt is added to water, density of the solution will increase.
Number of correct statement is
a) 0
b) 1
c) 2
d) None of the mentioned
Answer
Answer: c [Reason:] Specific density is the ratio of two densities and when salt is added to water, a high density material is added so finally the density rises .
8. An object has specific gravity greater than 1. When it is kept on the surface of water, the object would
a) Sink
b) Float
c) Half part of the object floats
d) None of the mentioned
Answer
Answer: a [Reason:] With respect to water, the specific gravity is greater than one so the density of the object is more than that of water.
9-10. A rectangular block measures 2 x 5 x 8 centimetres. Mass of the block is 66 grams.
9. Density of the block is?
a) 0.625 gm/cm3
b) 0.825 gm/cm3
c) 1.125 gm/cm3
d) 2.225 gm/cm3
Answer
Answer: b [Reason:] Density is the ratio of mass and volume of the block.
10. Specific gravity is?
a) 0.625
b) 0.825
c) 1.125
d) 2.225
Answer
Answer: b [Reason:] Specific density = 0.825/1 = 0.825, where the density of water is 1 gm/cm3.
Set 4
1-3. Volume representation of a real gas is given by the equation
V = aT + bTP + cTPn Litre
Where: a,b,c are constants and T, P, n are temperature, pressure and number of moles respectively.
If the equation is dimensionally consistent then
1. What is the unit of a?
a) L/K
b) L/(K)(atm)
c) L/(K)(atm)(mole)
d) None of the mentioned
Answer
Answer: a [Reason:] Unit of a = unit of V / unit of T.
2. What is the unit of b?
a) L/K
b) L/(K)(atm)
c) L/(K)(atm)(mole)
d) None of the mentioned
Answer
Answer: b [Reason:] Unit of b = unit of V / (unit of T)(Unit of P).
3. What is the unit of c?
a) L/K
b) L/(K)(atm)
c) L/(K)(atm)(mole)
d) None of the mentioned
Answer
Answer: c [Reason:] Unit of c = unit of V / (unit of T)(Unit of P)(unit of n).
4. A relation of distance with time is given as d = 25(1-exp(-0.021t)) µm where d is in microns and t in seconds. What is the relation if d is in inches and t in minutes?
a) 25(1-exp(-0.021t)) in
b) 1.64(1-exp(0.26t)) in
c) 3.84(1-exp(0.26t)) in
d) 9.84(1-exp(1.26t)) in
Answer
Answer: d [Reason:] Each term in an equation must have same dimensions and units as every other term to which it is added or subtracted or equated.
5. Given relation are-
A. T = 4π[l/g]0.5
Where: l- length, g- gravity, T- time period of a pendulum
B. ∆P = v2*(L/D)
Where: ∆P- Pressure drop, L- length of pipe and D- Diameter of the pipe
Having dimensional consistency
a) 1
b) 2
c) Both 1 and 2
d) Neither 1 nor 2
Answer
Answer: a [Reason:] Units of left hand-side of the equation match the units of the right-hand side, the equation is dimensionally consistent.
6. For dimensional consistency each term in an equation must have the same net dimension and unit as every other term to which it is added or subtracted or equated.
The above given statement is
a) False
b) True
c) Not mandatory
d) None of the mentioned
Answer
Answer: b [Reason:] For dimensional consistency each term in an equation must have the same net dimension and unit as every other term to which it is added or subtracted or equated.
7. A collection of variables or parameters that has no net dimensions or units are called
a) Dimensionless group
b) Dimensional group
c) Derived dimension group
d) None of the mentioned
Answer
Answer: a [Reason:] A collection of variables or parameters that has no net dimensions or units are called dimensionless group.
8. Find out the dimensionless quantity
a) Pressure
b) Reynolds number
c) Viscosity
d) Density
Answer
Answer: b [Reason:] Reynolds number has no dimension or units.
9-10. Pressure relation of a real gas is given by the equation P = Aexp(B/T) atm. Where A and B are constants and T is the temperature of the gas.
9. What is the unit of A?
a) atm
b) K
c) 1/atm
d) 1/K
Answer
Answer: a [Reason:] The unit of A is same as the unit of pressure.
10. What is the unit of B?
a) atm
b) K
c) 1/atm
d) 1/K
Answer
Answer: b [Reason:] The unit of B is same as the unit of temperature.
Set 5
1-3. For the given hydro-cracking problem, mole percent of products C4H10, C3H8 and C2H6 are 20.2%, 58.4% and 21.4% respectively.
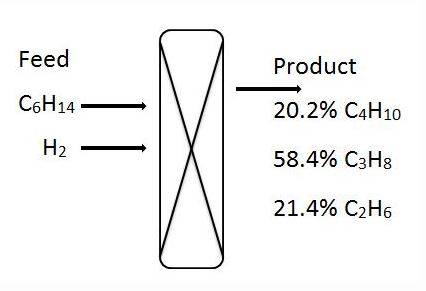
1. What is the mole percent of C6H14 in the feed?
a) 40.2
b) 50.2
c) 60.2
d) 70.2
Answer
Answer: c [Reason:] Element material balance for each element.
2. What is the mole percent of H2 in the feed?
a) 59.2
b) 49.2
c) 39.2
d) 29.9
Answer
Answer: b [Reason:] Element material balance for each element.
3. Molar ratio of H2 consumed to Hexane reacted in the process?
a) 1.2
b) 1.4
c) 1.6
d) 1.8
Answer
Answer: a [Reason:] Element material balance for each element.
4-6. For producing 40 % Methane, 20% Ethane and 40% Butane, a reactor is feed with Pentane and Hydrogen.
4. What is the mole fraction of Hydrogen in the feed?
a) 0.284
b) 0.484
c) 0.684
d) 0.884
Answer
Answer: c [Reason:] Element material balance for each element.
5. What is the mole fraction of Pentane in the feed?
a) 0.315
b) 0.215
c) 0.115
d) None of the mentioned
Answer
Answer: a [Reason:] Element material balance for each element.
6. What is the ratio of Hydrogen to the Pentane in the feed?
a) 1.167
b) 2.167
c) 3.167
d) 4.167
Answer
Answer: b [Reason:] Ratio = 104/48 = 2.167.
7-10. C3H8 is burned in air with a composition of 40 moles and 100 moles.
7. What is the number of moles of CO2 in the product?
a) 12.6
b) 10.6
c) 8.6
d) 4.6
Answer
Answer: a [Reason:] Moles of CO2 = Extent of the reaction*3.
8. What is the number of moles of H2O in the product?
a) 4.8
b) 8.8
c) 16.8
d) 32.8
Answer
Answer: c [Reason:] Moles of H2O = Extent of the reaction*2.
9. What is the number of moles of C3H8 in the product?
a) 25.8
b) 35.8
c) 45.8
d) 55.8
Answer
Answer: b [Reason:] Moles of C3H8 = 40-Extent of the reaction.
10. How many moles of O2 are required for the complete combustion of C3H8?
a) 160.48
b) 170.48
c) 180.48
d) 190.48
Answer
Answer: d [Reason:] Suppose x moles are required then 0.21x = 48.
