Multiple choice question for engineering
Set 1
1. Which of the following is the most sensitive of the spectral methods?
a) Absorption spectroscopy
b) Mass spectroscopy
c) Flame emission spectroscopy
d) Atomic emission spectroscopy
Answer
Answer: b [Reason:] Mass spectroscopy is the most sensitive of the spectral methods. It permits direct introduction of a gas effluent’s stream.
2. Which of the following is the disadvantage of gas chromatography?
a) It is not a good method
b) It cannot be used for qualitative analysis
c) It cannot be used for separation of volatile components
d) It does not provide direct identification
Answer
Answer: d [Reason:] Gas chromatography is a very good method for separation of components of a mixture. It does not provide direct identification.
3. Cold tray is provided for liquid N2 or CO2 but is used only for extremely small samples.
a) True
b) False
Answer
Answer: a [Reason:] Cold tray is provided for liquid N2 or CO2. It is used only for mass spectral analysis of very small samples.
4. Which of the following is most often used in the chromatograph in gas chromatograph MS?
a) Curvette
b) Paper support
c) Capillary tube
d) Flask
Answer
Answer: c [Reason:] Capillary tube is most often used in the chromatograph in gas chromatograph MS. This is because low pressure has to be maintained.
5. GC- MS has been developed for which of the following systems?
a) Packed column
b) Open tubular column
c) Capillary column
d) Porous layer column
Answer
Answer: a [Reason:] GC- MS has been developed for packed column system. These allow for analyte molecules to be dynamically extracted from the carrier gas stream.
6. Which of the following problems occur when combining gas chromatography and mass spectroscopy?
a) Difference in operating pressures
b) Reduction in sensitivity
c) Direct identification is not possible
d) It does not permit direct introduction of the effluent
Answer
Answer: The problem that occurs when combining gas chromatography and mass
spectroscopy is the difference in operating pressures. The pressure at the exit of the GC column is atmospheric.
7. The carrier gas with low molecular weight would diffuse at a higher rate than the higher molecular weight sample.
a) True
b) False
Answer
Answer: a [Reason:] The carrier gas with low molecular weight would diffuse at a higher rate than the higher molecular weight sample. The carrier gas would diffuse away from the line of flow.
8. Which of the following is the type of separator used in commercial GC-MS systems?
a) Jet type molecular separator
b) Porous tube
c) Teflon tube
d) Flow type separator
Answer
Answer: a [Reason:] Jet type molecular separator is the type of separator used in commercial GC-MS systems. Other methods are porous tube and Teflon tube.
9. The system for measurement of ion intensity in GS-MS system consists of which of the following?
a) Electrometer
b) Ion meter
c) Ion transducer
d) Intensity meter
Answer
Answer: a [Reason:] The system for measurement of ion intensity in GS-MS system consists of electrometer. It feeds a direct writing recorder.
10. The system for measurement of ion intensity in GS-MS system consists of which of the following?
a) Band pass amplifier
b) Narrow band amplifier
c) Wide band amplifier
d) Low pass amplifier
Answer
Answer: c [Reason:] The system for measurement of ion intensity in GS-MS system consists of a wide band amplifier. It feeds a direct writing recorder.
Set 2
1. Which of the following is not the characteristic of a reference electrode?
a) It must have a known output potential
b) It must have a constant output potential
c) Its output potential is dependent on the composition of the solution
d) It is employed in conjunction with the indicator or working electrode
Answer
Answer: c [Reason:] The output potential of a reference electrode must be insensitive to the composition of the solution.
2. Why is Standard hydrogen electrode called as the primary reference electrode?
a) It has a known output potential
b) It has a constant output potential
c) Its output potential is independent of the composition of the solution
d) Its output potential is zero volts
Answer
Answer: d [Reason:] Standard hydrogen electrode is called the primary reference electrode as its output potential is zero volts. It is employed in conjunction with the indicator or working electrode.
3. Which of the following is the simple and most convenient hydrogen electrode?
a) Pascal Hydrogen electrode
b) Bourne Hydrogen electrode
c) Hilderbant Hydrogen electrode
d) West Hydrogen electrode
Answer
Answer: c [Reason:] The hydrogen electrode given by Hilderbant is the simple and most convenient hydrogen electrode. A number of hydrogen electrodes are available.
4. Which of the following is not the disadvantage of hydrogen electrode?
a) Platinum can be easily poisoned
b) Presence of oxidising agents alters the potential
c) It gives salt error
d) H2 gas at 1 atmospheric pressure is difficult to set up and transport
Answer
Answer: c [Reason:] Hydrogen electrode does not give salt error. A number of hydrogen electrodes are available.
5. In Hydrogen electrode, the electrode is placed in a solution of ____ M Hcl. Fill in the blank.
a) 0.5
b) 1
c) 2
d) 3
Answer
Answer: b [Reason:] In Hydrogen electrode, the electrode is placed in a solution of 1M Hcl. H2 gas at 1 atm pressure is passed through the side arm in such a way that the platinum is half immersed in Hcl.
6. Hydrogen electrode which is the reference electrode can be used as which of the following?
a) Anode only
b) Cathode only
c) Anode or Cathode
d) Salt bridge
Answer
Answer: c [Reason:] Hydrogen electrode which is the reference electrode can be used as the anode or the cathode. It depends on the half-cell to which it is coupled.
7. If hydrogen electrode acts as cathode, hydrogen is reduced.
a) True
b) False
Answer
Answer: a [Reason:] If hydrogen electrode acts as cathode, hydrogen is reduced. If hydrogen electrode acts as anode, hydrogen is oxidised.
8. Given below is a diagram of hydrogen electrode. Identify the unmarked component.
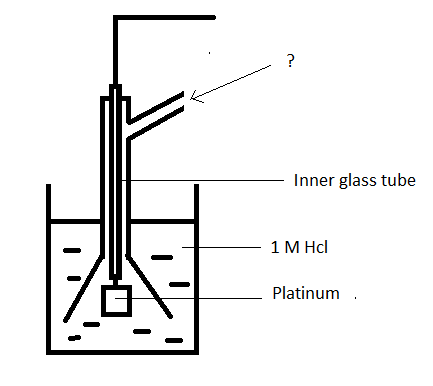
a) Hydrogen at 1 atm
b) Hydrogen at 10 atm
c) Helium at 1 atm
d) Helium at 10 atm
Answer
Answer: a [Reason:] Hydrogen at 1 atm is sent through the side tube. The electrode is placed in a solution of 1M Hcl.
9. The composition of glass membrane in glass electrode cannot have which of the following?
a) Sodium silicate
b) Calcium silicate
c) Lithium silicate
d) Barium silicate
Answer
Answer: d [Reason:] Glass electrode consists of either sodium or calcium silicate or lithium silicates containing glass membrane. It has lanthanum and barium ions added to the membrane.
10. Which of the following is the purpose of added membranes in the glass membrane of the glass electrode?
a) They act as tightners
b) They act as filters
c) They act as conditioners
d) They act as collectors
Answer
Answer: a [Reason:] The ions in the added membranes act as tightners. They reduce the mobility of sodium ion.
11. Which of the following cannot form the inner reference electrode in glass electrodes?
a) Silver electrode
b) Copper electrode
c) Calomel electrode
d) Silver chloride electrode
Answer
Answer: b [Reason:] Copper electrode cannot form the inner reference electrode in glass electrodes. Inner reference electrode is immersed in a buffer solution.
12. The pH response of glass electrode is limited entirely to the area of the special glass membrane bulb.
a) True
b) False
Answer
Answer: a [Reason:] The pH response of glass electrode is limited entirely to the area of the special glass membrane bulb. The response of the electrode is independent of the depth of immersion.
13. Which of the following is not the advantage of glass electrodes?
a) It gives accurate results for high as well as low pH values
b) It is simple to operate
c) It has no salt error
d) Modern electrodes can withstand severe treatment
Answer
Answer: a [Reason:] It gives accurate results for low pH values only ie. from 0 to 9. For high pH values, the glass becomes responsive to sodium and other cations.
14. Which of the following is not the disadvantage of glass electrodes?
a) Poor readings are obtained in buffered or unbuffered solutions
b) The electrode must be washed thoroughly with distilled water to obtain proper results
c) Materials suspended on glass should be wiped out neatly to obtain proper results
d) It is affected by oxidation reduction potentials in the solution
Answer
Answer: d [Reason:] It is affected by oxidation reduction potentials in the solution. It is an advantage of the glass electrode.
15. Given below is the diagram of glass electrode. Identify the unmarked component.
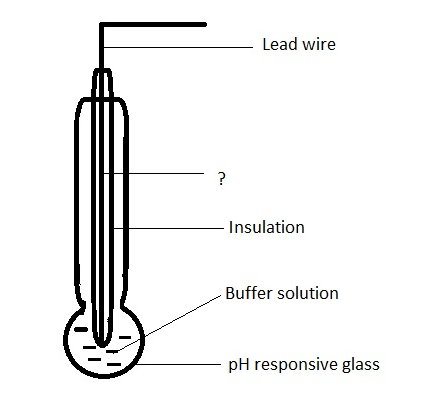
a) Platinum leads
b) Silver wire coated with silver chloride
c) Copper wire
d) Platinum reference electrode
Answer
Answer: b [Reason:] The unmarked component is silver wire coated with silver chloride. It forms the inner reference electrode.
Set 3
1. In ion spectroscopy, the positive ions are focussed on the sample at which of the following angles?
a) 20o
b) 30o
c) 45o
d) 90o
Answer
Answer: c [Reason:] In ion spectroscopy, the positive ions are focussed on the sample at 45o. Ions are formed by bombardment.
2. Only those electrons which are in a selected small solid angle are received in the ________ electrostatic analyser.
a) 100o
b) 127o
c) 180o
d) 263o
Answer
Answer: b [Reason:] Only those electrons which are in a selected small solid angle are received in the 127o electrostatic analyser. Ions are scattered in all directions.
3. Which of the following can be used as the detector in ion spectroscopy?
a) Faraday cup collector
b) Channel electron multiplier
c) Micro-channel plate
d) Flame ionization detector
Answer
Answer: b [Reason:] Channel electron multiplier can be used as the detector in ion spectroscopy. Solid state detector can also be used.
4. In order to obtain ISS spectra, the backscattered primary ions are sampled by which of the following?
a) Faraday cup analyser
b) Photographic analyser
c) Micro-channel analyser
d) Cylindrical mirror analyser
Answer
Answer: d [Reason:] In order to obtain ISS spectra, the backscattered primary ions are sampled by cylindrical mirror analyser. Their kinetic energies are then measured.
5. Which of the following is the energy after collision with a surface atom for a scattering angle of 90o when Eo is the energy of the incident ion, M1 is the mass of the incident ion and M2 is the mass of the target surface?
a) Eo(M2-M1)/(M2+M1)
a) Eo(M2+M1)/(M2-M1)
a) Eo(M2×M1)/(M2+M1)
a) Eo(M2-M1)/(M2×M1)
Answer
Answer: a [Reason:] Eo(M2-M1)/(M2+M1) is the energy after collision with a surface atom for a scattering angle of 90o. But, this is valid only when M1<M2.
6. It is advantageous to use ions from a variety of gases.
a) True
b) False
Answer
Answer: a [Reason:] It is advantageous to use ions from a variety of gases. Few gases are chosen to avoid side effects.
7. Which of the following gases is not often used in ion spectroscopy?
a) Helium
b) Argon
c) Nitrogen
d) Neon
Answer
Answer: c [Reason:] Helium, argon and inert gases are usually chosen for ion spectroscopy. These gases are chosen to avoid side effects.
8. ISS is sensitive to every element heavier than which of the following?
a) Helium
b) Hydrogen
c) Nitrogen
d) Neon
Answer
Answer: a [Reason:] ISS is sensitive to every element heavier than helium. The lightest isotope used as primary ion is that of helium.
9. Which of the following is the lightest isotope used as a primary ion?
a) He
b) 2He
c) 3He
d) 3H
Answer
Answer: c [Reason:] 3He is the lightest isotope used as a primary ion. Though hydrogen is lightest it is not used.
10. The specificity will vary depending on the scattering gas used.
a) True
b) False
Answer
Answer: a [Reason:] The specificity will vary depending on the scattering gas used. Only few gases are preferred to avoid side effects.
11. ISS is less sensitive than which of the following?
a) SIMS
b) Auger
c) ESCA
d) AES
Answer
Answer: a [Reason:] ISS is less sensitive than SIMS. It is more sensitive than Auger or ESCA.
12. Which of the following is the detection limit of ISS for monolayer?
a) 10 -1 %
b) 10 -2 %
c) 10 -3 %
d) 10 -4 %
Answer
Answer: c [Reason:] 10-3% of the monolayer is the detection limit of ISS. It can be effectively used for depth profiling.
13. After passing through the pre-filter, ions are passed through which of the following?
a) Quadrupole mass spectrometer
b) Time of flight mass spectrometer
c) Radiofrequency mass spectrometer
d) Magnetic deflection mass spectrometer
Answer
Answer: a [Reason:] After passing through the pre-filter, ions are passed through quadrupole mass spectrometer. Pre-filter is a discriminator.
14. Which of the following is the range of quadrupole mass spectrometer used in SIMS?
a) 100-200 amu
b) 200-300 amu
c) 500-1000 amu
d) 200-500 amu
Answer
Answer: c [Reason:] 500-1000 amu is the range of quadrupole mass spectrometer used in SIMS. SIMS shows good specificity.
15. Which of the following is the resolution of quadrupole mass spectrometer used in SIMS?
a) 1 amu
b) 2 amu
c) 5 amu
d) 3 amu
Answer
Answer: a [Reason:] 1 amu is the resolution of quadrupole mass spectrometer used in SIMS. SIMS shows good specificity.
Set 4
1. Which of the following is used as detector crystal in ESR spectrometer?
a) Silicon rectifier
b) Silicon tungsten rectifier
c) Silicon boron rectifier
d) Silicon quartz rectifier
Answer
Answer: b [Reason:] Silicon tungsten rectifier is used as detector crystal in ESR spectrometer. This is the commonly used detector.
2. After detection, the resulting signal will have which of the following frequencies?
a) 1 kHz
b) 10 kHz
c) 100 kHz
d) 1000 kHz
Answer
Answer: c [Reason:] After detection, the resulting signal will have 100 kHz. It contains ESR information.
3. Which of the following is the modulation amplitude range of ESR spectrometer?
a) 1mG to 10G
b) 2mG to 15G
c) 3mG to 20G
d) 5mG to 40G
Answer
Answer: d [Reason:] 5mG to 40G is the modulation amplitude range of ESR spectrometer. It is applied in steps.
4. In which of the following ranges will the receiver time constant of the ESR spectrometer lie?
a) 0 to 10 s
b) 0.3 to 1 s
c) 0.03 to 10 s
d) 0.003 to 100 s
Answer
Answer: d [Reason:] The receiver time constant of the ESR spectrometer lies from 0.003 to 100 s. It is one of the important parameters of ESR spectrometer.
5. Which of the following is the oscilloscope field width of the ESR spectrometer?
a) 0.2 to 40 G
b) 0.1 to 20 G
c) 0 to 10 G
d) 0.01 to 20 G
Answer
Answer: a [Reason:] The oscilloscope field width of the ESR spectrometer lies between 0.2 to 40 G. it is one of the important parameters.
6. The magnet used in ESR spectrometer provides a magnetic field which can be varied from ________ to _______
a) 20 mgauss, 100mgauss
b) 200 mgauss, 20kgauss
c) 20 mgauss, 20kgauss
d) 100 mgauss, 10kgauss
Answer
Answer: b [Reason:] The magnet used in ESR spectrometer provides a magnetic field which can be varied from 200 mgauss to 20kgauss. It is calibrated in steps.
7. Which of the following sensor is used in ESR spectrometer?
a) Hall-effect sensor
b) Load cell
c) Strain gauge
d) Bourdon gauge
Answer
Answer: a [Reason:] Hall-effect sensor is used in ESR spectrometer. It is given as an input to the summation circuit.
8. Which of the following oscillator is used as 1230 Hz oscillator?
a) Hartley oscillator
b) Crystal oscillator
c) RC oscillator
d) Wien bridge oscillator
Answer
Answer: d [Reason:] Wien bridge oscillator is used as 1230 Hz oscillator. The output level is internally regulated.
9. What does AFC stand for?
a) Auto frequency correct
b) Automated frequency correct
c) Automatic frequency control
d) Automatic frequency circuit
Answer
Answer: c [Reason:] AFC stands for automatic frequency control. A 70 kHz AFC is used.
10. Klystron requires _______ for the klystron beam and up to _____ for the klystron reflector.
a) +400 V, -730 V
b) +650 V, -400 V
c) +200 V,-530 V
d) +180 V,-270 V
Answer
Answer: b [Reason:] Klystron requires +650 V for the klystron beam and up to -400 V for the klystron reflector. Klystron is also known as reflex oscillator.
11. The klystron body is insulated from ground by an insulating gas casket.
a) True
b) False
Answer
Answer: a [Reason:] The klystron body is insulated from ground by an insulating gas casket. It is placed between klystron flange and water cooling flange.
12. The 100 kHz oscillator acts as which of the following?
a) Transmitter
b) Receiver
c) Modulator
d) Transmitter and receiver
Answer
Answer: d [Reason:] The 100 kHz oscillator acts as the transmitter and receiver. It is crystal controlled.
13. Which of the following removes residual harmonics from the phase-detected signal?
a) High-pass filter
b) Low-pass filter
c) Band-pass filter
d) Chopper
Answer
Answer: b [Reason:] Low-pass filter removes residual harmonics from the phase-detected signal. It removes the residual 100 kHz if any.
14. The oscilloscope is used to display the klystron mode.
a) True
b) False
Answer
Answer: a [Reason:] The oscilloscope is used to display the klystron mode. It provides visual observation of rapidly changing signals.
15. In the sample cell, which of the following is selected to give maximum optical transmission in the UV-visible region?
a) Tungsten
b) Quartz
c) Phosphor
d) Potassium
Answer
Answer: b [Reason:] In the sample cell, quartz is selected to give maximum optical transmission in the UV-visible region. Aqueous sample cells are specially designed.
Set 5
1. In X-ray spectrometers, the specimen or the sample is placed after which of the following components?
a) X-ray tube
b) Monochromator
c) Collimator
d) Detector
Answer
Answer: a [Reason:] In X-ray spectrometers, the specimen or the sample is placed after the X-ray tube. The X-ray tube is the source of the X-ray.
2. Which of the following components are used to generate X-rays?
a) Meyer tube
b) West tube
c) Anger tube
d) Coolidge tube
Answer
Answer: d [Reason:] Coolidge tube is used to generate X-rays. It the source of X-rays. Coolidge tube requires stabilised current and high voltage.
3. Using which of the following components is the generated x-rays focussed upon the specimen?
a) X-ray tube
b) Monochromator
c) Collimator
d) Detector
Answer
Answer: c [Reason:] Collimator is used to focus the generated x-rays upon the specimen. The collimator is in between the specimen under analysis and the Coolidge tube.
4. The cathode in the Coolidge tube is made of which of the following elements?
a) Quartz
b) Iron
c) Tungsten
d) Barium
Answer
Answer: c [Reason:] The cathode in the Coolidge tube is made of tungsten. The anode is made of copper.
5. The cathode in the Coolidge tube is kept in an inclined manner.
a) True
b) False
Answer
Answer: b [Reason:] The anode in the Coolidge tube is kept in an inclined manner. The anode is made of copper.
6. Which of the following is not a target metal used in Coolidge tube?
a) Rhodium
b) Cobalt
c) Gold
d) Silver
Answer
Answer: c [Reason:] Gold is not used as a target metal in Coolidge tube. The other target metals are copper, molybdenum and chromium.
7. How can the resolution of the collimator be increased?
a) By reducing the separation between the metal plates of the collimator
b) By increasing the separation between the metal plates of the collimator
c) By increasing the number of metal plates
d) By decreasing the number of metal plates
Answer
Answer: a [Reason:] The resolution of the collimator can be increased by reducing the separation between the metal plates of the collimator. Collimator has a series of closely spaced parallel metal plates.
8. When x-rays emitted from molybdenum are allowed to pass through a zirconium filter, which of the following occurs?
a) It absorbs radiation of shorter wavelength
b) It absorbs radiation of longer wavelength
c) It allows radiation of shorter wavelength to pass through
d) It allows radiation in a particular band to pass through
Answer
Answer: b [Reason:] When x-rays emitted from molybdenum are allowed to pass through a zirconium filter, it absorbs radiation of shorter wavelength. It allows radiation of stronger wavelength to pass through.
9. When compared to filters, monochromators provide much signal to noise ratio.
a) True
b) False
Answer
Answer: a [Reason:] When compared to filters, monochromators provide much signal to noise ratio. Monochromators are used for removal of unwanted wavelengths.
10. Which of the following crystals are not suited for x-ray grating?
a) Topaz
b) Lithium fluoride
c) Calcium fluoride
d) Sodium fluoride
Answer
Answer: d [Reason:] The crystal which is not suited for x-ray grating is sodium fluoride. Other crystals which are suitable for x-ray grating are gypsum and sodium chloride.
11. Given below is the block diagram of X-ray spectrometer. Identify the unmarked component.
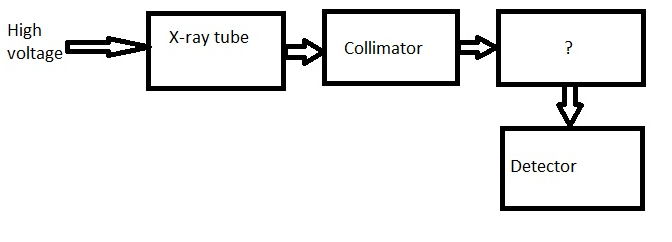
a) Filter
b) Monochromator
c) Specimen
d) Amplifier
Answer
Answer: c [Reason:] The unmarked component is specimen. It is the sample under analysis.
12. The x-rays generated come out of the Coolidge tube through which of the following?
a) Beryllium window
b) Tungsten window
c) Collimator
d) Target material
Answer
Answer: a [Reason:] The x-rays generated come out of the Coolidge tube through beryllium window. Some energy is lost as heat.
