Multiple choice question for engineering
Set 1
1. Which of the following is the disadvantage of reciprocating pump used in liquid chromatography?
a) Produces pulsed flow
b) Corrosive components
c) Does not have small hold-up value
d) Does not have moderate flow rate
Answer
Answer: a [Reason:] The disadvantage of reciprocating pump used in liquid chromatography is that it produces pulsed flow. Therefore, the flow must be damped before it affects the column.
2. Which of the following is not a disadvantage of Pneumatic pumps used in liquid chromatography?
a) Pulsed output
b) Dependent on solvent viscosity
c) Dependent on back pressure
d) Inconvenient for solvent gradient elution
Answer
Answer: a [Reason:] Pneumatic pumps provide pulse free output. They are inexpensive.
3. Which of the following is not a desired characteristic of pulse dampers or flow smootheners used in liquid chromatography?
a) Easy mobile phase change over
b) Constant flow must be maintained
c) Should be effective at low system pressure
d) Maximal dead volume
Answer
Answer: d [Reason:] Pulse dampers are used to avoid variations in flow. They must have minimal dead volume.
4. Which of the following will improve the efficiency of separation process in liquid chromatography?
a) Increase in sample size, increase in column diameter
b) Reduction in sample size, increase in column diameter
c) Increase in sample size, reduction in column diameter
d) Reduction in sample size, reduction in column diameter
Answer
Answer: d [Reason:] Reduction in sample size and reduction in column diameter will improve the efficiency of separation process in liquid chromatography. The effect of uneven flow will also be reduced.
5. Which of the following are the practical problems that arise due to the decrease in column diameter?
a) Requirement of large particle size and high pressure drop
b) Requirement of large particle size and low pressure drop
c) Requirement of small particle size and high pressure drop
d) Requirement of small particle size and low pressure drop
Answer
Answer: c [Reason:] The practical problems that arise due to decrease in column diameter are requirement of small particle size and high pressure drop. But, it increases column efficiency.
6. Which of the following is not true about guard column used in liquid chromatography?
a) It filters particles that clog the separation column
b) It extends the lifetime of separation column
c) It allows particles that cause precipitation upon contact with stationary or mobile phase
d) The size of packing varies with the type of protection needed
Answer
Answer: c [Reason:] Guard column removes particles that cause precipitation upon contact with stationary or mobile phase. It is placed before the separation column.
7. Which of the following columns are not used in liquid or high performance liquid chromatography?
a) Analytical column
b) Separation column
c) Guard column
d) Capillary column
Answer
Answer: d [Reason:] Capillary column is used in gas chromatography. Analytical column is also known as separation column.
8. Which of the following is not a Column-type Liquid chromatography?
a) Gel permeation
b) Ion exchange
c) Liquid-solid
d) Paper
Answer
Answer: d [Reason:] Paper chromatography is not a column-type chromatography. It makes use of strips of hollow cylinders of filter paper.
9. Which of the following is not true about radial compression column when compared to standard separation column?
a) Internal diameter decreases
b) Overall operating pressure decreases
c) Analysis time decreases
d) Solvent flow increases
Answer
Answer: a [Reason:] Radial compression columns have a wider diameter than standard separation column. The cartridges used are of low cost.
10. Which of the following is not true about narrow bore column when compared to standard columns?
a) Internal diameter decreases
b) Volumetric flow decreases
c) Solvent cost is saved
d) Detector response time increases
Answer
Answer: d [Reason:] Detector response time increases in narrow bore columns. High purity solvents must be used in these columns.
11. Which of the following types of liquid chromatography uses immobilized biochemical as stationary phase?
a) Ion exchange chromatography
b) Exclusion chromatography
c) Affinity chromatography
d) Gel permeation chromatography
Answer
Answer: c [Reason:] In Affinity chromatography, immobilized biochemical is used as stationary phase. It is used to separate one or few solutes from hundreds of unretained solutes.
Set 2
1. Which of the following does not occur in Hartman and Braun oxygen analyzer based on magnetic wind?
a) Oxygen is attracted to magnetic field
b) Oxygen becomes less paramagnetic due to rise in temperature
c) Resistance change in a resistance connected to the Wheatstone bridge occur
d) Wheatstone Bridge becomes balanced due to temperature change
Answer
Answer: d [Reason:] In Hartman and Braun oxygen analyzer, the magnetic wind cools the heated winding. As a result, the bridge becomes unbalanced.
2. Which of the following is not a cause of error that could occur in Hartman and Braun oxygen analyzer based on magnetic wind?
a) The filament temperature could be affected by changes in thermal conductivity of the carrier gas
b) Hydrocarbons in sample stream may react on the heated filaments
c) Gravitation chimney flow effect lead to errors when cross tube is horizontal
d) Combustible gases may react on the heated filaments
Answer
Answer: c [Reason:] Error occurs when cross tube is vertical. When cross tube is horizontal, errors due to gravitation chimney flow effect is avoided.
3. Hydrocarbons need to be removed from the sample gas to avoid errors in Hartman and Braun oxygen analyzer.
a) True
b) False
Answer
Answer: a [Reason:] Hydrocarbons need to be removed from the sample gas to avoid errors. This is because they may react on the heated filaments and lead to change in temperature and resistance.
4. Magnetic wind analyzers are also known as which of the following?
a) Thermo-magnetic analyzers
b) Electro-magnetic analyzers
c) Thermo-analytical analyzers
d) Hot wire analyzers
Answer
Answer: a [Reason:] Magnetic wind analyzers are also known as Thermo-magnetic analyzers. In Europe, it is known as Magnetic wind analyzers. In United States, it is known as Thermo-magnetic analyzers.
5. In OXYMAT-M oxygen analyser which of the following is usually used as reference gas?
a) Hydrogen
b) Nitrogen
c) Helium
d) Ambient air
Answer
Answer: d [Reason:] In OXYMAT-M oxygen analyser, ambient air is usually used as reference gas. Its measuring procedure is based on pressure difference which develops between two gases having different oxygen concentration in the magnetic field.
6. Which of the following is used in magnetic wind instruments to determine the amount of oxygen?
a) Change in resistance
b) Change in capacitance
c) Change in inductance
d) Change in concentration
Answer
Answer: a [Reason:] In magnetic wind instruments to determine the amount of oxygen, change in temperature leads to a change in resistance. This change in resistance is measured to determine the amount of oxygen.
7. Which of the following concepts are not used in magnetic wind instruments?
a) Change in resistance due to cooling by magnetic wind
b) Displacement of membrane by magnetised oxygen
c) Difference in pressure difference between two gases having varying concentration of oxygen
d) By measuring temperature using thermometer
Answer
Answer: d [Reason:] Temperature is not measured in magnetic wind instruments. The cooling effect of magnetic wind causes a change in resistance which is measured.
8. How are the hydrocarbons in the samples removed in magnetic wind instruments?
a) By using a cold trap
b) By using a filter
c) By using a semipermeable membrane
d) By using magnetic effect
Answer
Answer: a [Reason:] The hydrocarbons in the samples are removed in magnetic wind instruments by using a cold trap. It is removed to avoid combustion.
9. Which of the following bridges are used in magnetic wind instruments?
a) Wheatstone bridge
b) Kelvin’s bridge
c) Anderson’s bridge
d) Schering’s bridge
Answer
Answer: a [Reason:] Wheatstone bridge is generally used in magnetic wind instruments. The change in resistance due to cooling effect is measured using Wheatstone bridge.
10. The extremely high inertia of the thin membrane in the oxygen sensor allows it to rapidly attain equilibrium.
a) True
b) False
Answer
Answer: b [Reason:] The extremely low inertia of the thin membrane in the oxygen sensor allows it to attain equilibrium. It reaches equilibrium state in 0.25 to 0.5 seconds.
Set 3
1. Nitrogen dioxide can be detected with a paramagnetic analyser.
a) True
b) False
Answer
Answer: a [Reason:] Nitrogen dioxide can be detected with a paramagnetic analyser. This is because nitrogen has paramagnetic property ie. it is attracted by magnetic fields.
2. Nitrogen oxide cannot be directly analysed using UV and Visible analyzers due to which of the following reasons?
a) Less accuracy
b) Very low range
c) It leads to contamination of the sample
d) It is transparent in UV visible regions
Answer
Answer: d [Reason:] Nitrogen oxide cannot be directly analysed using UV and Visible analyzers because it is transparent in UV visible regions. Hence, it is converted into nitrogen dioxide and then analysed.
3. How is NO converted to NO2 for analysis in UV and Visible analyzers?
a) Treating sample gas with pressurized oxygen
b) Treating sample gas with ozone
c) Treating sample gas with oxygen at low pressure
d) Treating sample gas with water at high pressure
Answer
Answer: a [Reason:] NO is converted into NO2 for analysis in UV and Visible analyzers by treating sample gas with pressurized oxygen. This is because direct measurement of NO is not possible using UV and Visible analyzers.
4. How is NO converted to NO2 for analysis in Chemiluminescent analyser?
a) Treating sample gas with pressurized oxygen
b) Treating sample gas with ozone
c) Treating sample gas with oxygen at low pressure
d) Treating sample gas with water at high pressure
Answer
Answer: b [Reason:] NO is converted to NO2 for analysis in Chemiluminescent analyser by treating sample gas with ozone.
NO + O3 —>NO2 + O2
5. During analysis of NO2 using Chemiluminescent analyser, why is NO2 not made to react with ozone directly?
a) Less accuracy
b) It is a slow process
c) It leads to contamination of the sample
d) It does not produce luminescence
Answer
Answer: b [Reason:] During analysis of NO2 using Chemiluminescent analyser, NO2 is not made to react with ozone directly because it is a slow process. Hence, it is converted to NO using catalytic reactions or converters.
6. The block diagram of series mode analyser of NO and NO2 is given below. Identify the unmarked procedure.
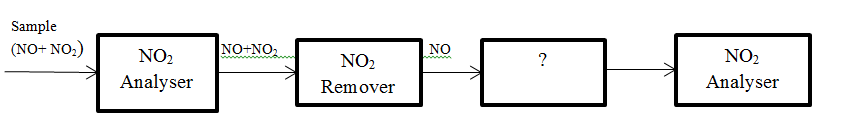
a) Oxidiser
b) Reducer
c) Filter
d) NO Analyser
Answer
Answer: a [Reason:] The unmarked procedure is NO to NO2 oxidiser. NO2 is then analysed by NO2 analyser.
7. The block diagram for parallel mode analyser of NO and NO2 is given below. Identify the unmarked block in the diagram.
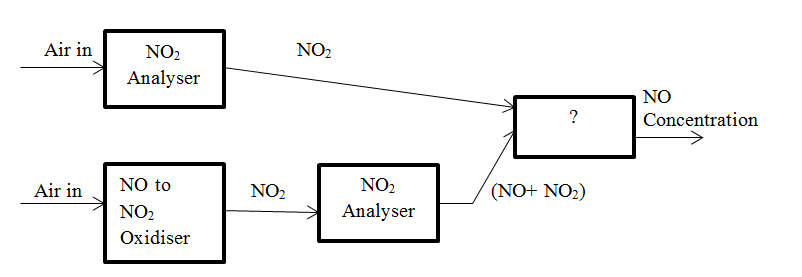
a) NO Analyser
b) NO2 to NO reduction
c) NO2 remover
d) Difference
Answer
Answer: d [Reason:] The unmarked block is Difference. Difference between the concentration of NO2 and NO and the concentration of NO2 alone is determined and the result gives the concentration of NO.
8. The instruments based on chemiluminescence maintain linearity in which of the following ranges?
a) 1ppb to 100ppb
b) 100ppb to 1000ppb
c) 1ppb to 1000ppb
d) 100ppb to 1000ppb
Answer
Answer: c [Reason:] The instruments based on chemiluminescence maintain linearity between 1ppb to 1000ppb. These are used to measure NO in exhaust gases in vehicles.
9. How can absorption be enhanced while determining NO concentration using CO laser?
a) By converting NO into NO2
b) By placing NO in a magnetic field
c) By using proper monochromators
d) By using choppers
Answer
Answer: b [Reason:] CO laser emits radiation that can be absorb NO. Absorption is enhanced by placing NO in a magnetic field.
10. Which of the following detectors are generally used for detection in NO analysis using CO laser?
a) Photomultiplier tube
b) Photovoltaic cell
c) Liquid nitrogen cooled Ge-Au element
d) Photo emissive tube
Answer
Answer: c [Reason:] Liquid nitrogen cooled Ge-Au element is used detection in NO analysis using CO laser. The signal amplitude varied with the concentration of NO.
11. Which of the following analyzers are used to measure trace amounts of nitrogen oxides in the stratosphere?
a) Chemiluminescence
b) CO laser method
c) Laser opto-acoustic spectroscopy
d) Colorimetry
Answer
Answer: c [Reason:] Laser opto-acoustic spectroscopy is used to measure trace amounts of nitrogen oxides in the stratosphere. It was developed for application in air pollution measurement.
12. A pink coloured dye complex is formed when air containing NO2 is passed in an absorbing solution consisting of __________ and diamine dissolved in acetic acid medium.
a) Sulphuric acid
b) Sulphonyl
c) Sulphonic acid
d) Sulphanilic acid
Answer
Answer: d [Reason:] A pink coloured dye complex is formed when air containing NO2 is passed in an absorbing solution consisting of sulphanilic acid and diamine dissolved in acetic acid medium. This is called Saltzman method.
13. In Laser Opto-acoustic spectroscopy, the IR beam excites the molecules to higher states. In which of the following ways do the molecules return to the ground state?
a) Collisional de-excitation
b) Random de-excitation
c) By spontaneous emission
d) By stimulated emission
Answer
Answer: a [Reason:] The molecules return to the ground state by collisional de-excitation. This results in increase in temperature.
14. To monitor oxides of nitrogen in stack effulents, the sample containing oxides of nitrogen is passed through a flask containing solution of H2O2 in sulphuric acid. Nitric acid is formed. The nitrate ions then react with phenol-disulphonic acid to produce blue colour.
a)True
b) False
Answer
Answer: b [Reason:] The nitrate ions react with phenol-disulphonic acid to produce yellow colour. This is measured colorimetrically.
Set 4
1. Which of the following steps takes place after injection of feed in Column chromatography?
a) Detection of components
b) Separation in the column
c) Elution from the column
d) Collection of eluted component
Answer
Answer: b [Reason:] The operation that occurs after the injection of feed is separation in the column. After that, elution from the column and detection of components takes place.
2. Components with strong attraction to the support move more slowly than components with weak attraction.
a) True
b) False
Answer
Answer: a [Reason:] Different components will adsorb to the stationary phase in varying degrees. Components with strong attraction to the support move more slowly than components with weak attraction.
3. What happens during the ‘elution from the column’ phase in chromatography?
a) Components with greatest affinity elute first
b) Components with least affinity elute first
c) Components elute in a random manner
d) Components elute according to their concentration in the mixture
Answer
Answer: b [Reason:] During the elution phase, different components elute at different times. Components with least affinity elute first.
4. In chromatogram, the position of peaks on the time axis can be used to determine which of the following?
a) Components of the sample
b) Amount of component in the sample
c) Column efficiency
d) Column resolution
Answer
Answer: a [Reason:] Chromatogram is a detector that responds to concentration solute and is placed at the end of the column. The position of peaks on the time axis can be used to determine components of the sample.
5. In chromatogram, the area under the peak can be used to determine which of the following?
a) Components of the sample
b) Amount of component in the sample
c) Column efficiency
d) Column resolution
Answer
Answer: b [Reason:] Chromatogram is a detector that responds to concentration solute and is placed at the end of the column. The area under the peak can be used to determine amount of component in the sample.
6. The stationary phase could be a viscous liquid coated over a surface of solid particles.
a) True
b) False
Answer
Answer: a [Reason:] The stationary phase could be a viscous liquid coated over a surface of solid particles. The solid particles can also be the stationary phase.
7. Given below is a diagram of separation of two components of a mixture in a chromatographic column. From the diagram, infer which component has lesser affinity to the stationary phase.
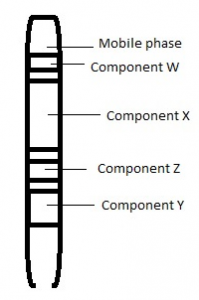
a) W
b) X
c) Y
d) Z
Answer
Answer: c [Reason:] Y will elute from the column first. Components with least affinity to the stationary phase will elute first.
8. Using Chromatogram as detector in Chromatography, a graph is obtained between ____________ and time.
a) Quantity
b) Density
c) Concentration
d) Specific gravity
Answer
Answer: c [Reason:] Using Chromatogram as detector in Chromatography, a graph is obtained between concentration and time. The detector is placed at the bottom of the column.
9. In older analytical methods, which of the following methods were used to allow movement of mobile phase?
a) Pumps
b) Pressure
c) Gravity
d) Blowing air into the column
Answer
Answer: c [Reason:] In older analytical methods, gravity facilitated the movement of mobile phase. The effect of capillary action was also employed to allow movement.
10. Given below is the diagram of ‘Process of chromatographic column’. Identify the unmarked component in the process.
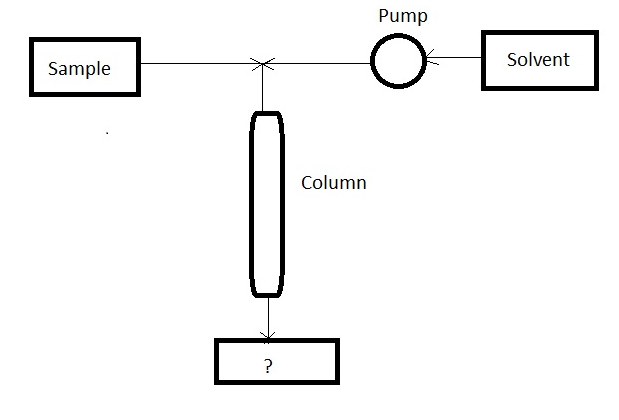
a) Reservoir
b) Collection tank
c) Microprocessor
d) Detector
Answer
Answer: d [Reason:] The unmarked component is detector. It is placed at the bottom of the column. It responds to solute concentration.
Set 5
1. Which of the following statements is false about single beam absorption instruments?
a) Tungsten bulb is used as source
b) Beam splitter is used to get parallel beam
c) Test tube is used as sample holder
d) Photovoltaic cell as detector
Answer
Answer: b [Reason:] Single beam instruments make use of one beam. Therefore, beam splitters are not required in single beam instruments.
2. Which of the following statement is false about double beam absorption instruments?
a) It is similar to single beam instruments except two beams are present
b) Tungsten bulb is used as source
c) Reference beam must have higher intensity than sample beam
d) Both the beams after they pass through respective samples are compared
Answer
Answer: c [Reason:] Reference beam cannot have higher intensity than sample beam. The beam is split into two beams of equal intensity.
3. Which of the following is not an application of colorimeter?
a) Paints
b) Inks
c) Cosmetics
d) Composition detection
Answer
Answer: d [Reason:] Colorimeter is not used to determine composition. Its application is paints, dyes, inks, cosmetics and plastics.
4. In photometers, the readings of the specimen are initially obtained in the form of which of the following parameters?
a) Transmittance
b) Absorption
c) Wavelengths
d) Volume
Answer
Answer: a [Reason:] In photometers, the reading is initially obtained in the form of transmittance as some radiation is absorbed by the sample and the rest of the beam is transmitted. This transmitted beam is measured by the detector.
5. Colorimeters are used in applications where great accuracy is required.
a) True
b) False
Answer
Answer: b [Reason:] Colorimeters are used in applications where great accuracy is not required. They are also known as photometers.
6. In the diagram of single beam photometer given below, identify the component that is not marked.
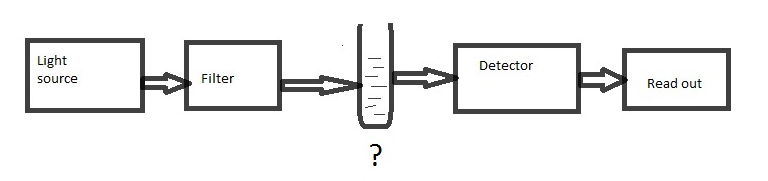
a) Monochromator
b) Absorption filter
c) Sample holder
d) Interference filter
Answer
Answer: c [Reason:] In single beam spectrophotometer, the beam passed through the sample which is held in the sample holder. The transmitted beam is measured by the detector.
7. Colorimeters are used to determine concentration of solutions.
a) True
b) False
Answer
Answer: a [Reason:] Colorimetry is the science of colour measurement. It is used to determine concentrations of solutions.
8. Which of the following is the purpose of balance indicator in double beam photometer or colorimeter?
a) Selects particular wavelength
b) Splits the wavelength selected into two equal beams
c) Detects and indicates the amount of light falling on it
d) Indicates the difference between the output of two photometers
Answer
Answer: d [Reason:] It compares the output of the two photometers obtained using two beams. It indicates the output.
9. Which of the following is the purpose of beam splitter in double beam photometer or colorimeter?
a) Splits beam into two equal intensity beams
b) Splits beam in such a way that sample beam has higher intensity
c) Splits beam in such a way that reference beam has higher intensity
d) Merge two equal intensity beams into single beam
Answer
Answer: a [Reason:] Beam splitter splits beam into two equal intensity beams. One beam passes through the sample and other through the reference.
10. Which of the following is a source used in spectroscopy?
a) LASER
b) Tube light
c) Sodium vapour lamp
d) Tungsten lamp
Answer
Answer: d [Reason:] Tungsten lamp is the source used in spectroscopy. It is the source used in UV, Visible spectroscopy.
